Antibody Drug Conjugates Contract Manufacturing Market Report
Published Date: 22 April 2026 | Report Code: antibody-drug-conjugates-contract-manufacturing
Antibody Drug Conjugates Contract Manufacturing Market Size, Share, Industry Trends and Forecast to 2033
This report provides comprehensive insights into the Antibody Drug Conjugates (ADCs) Contract Manufacturing market from 2023 to 2033, including market size, growth trends, regional analysis, and key players, making it essential for stakeholders in the biopharmaceutical industry.
Key Takeaways
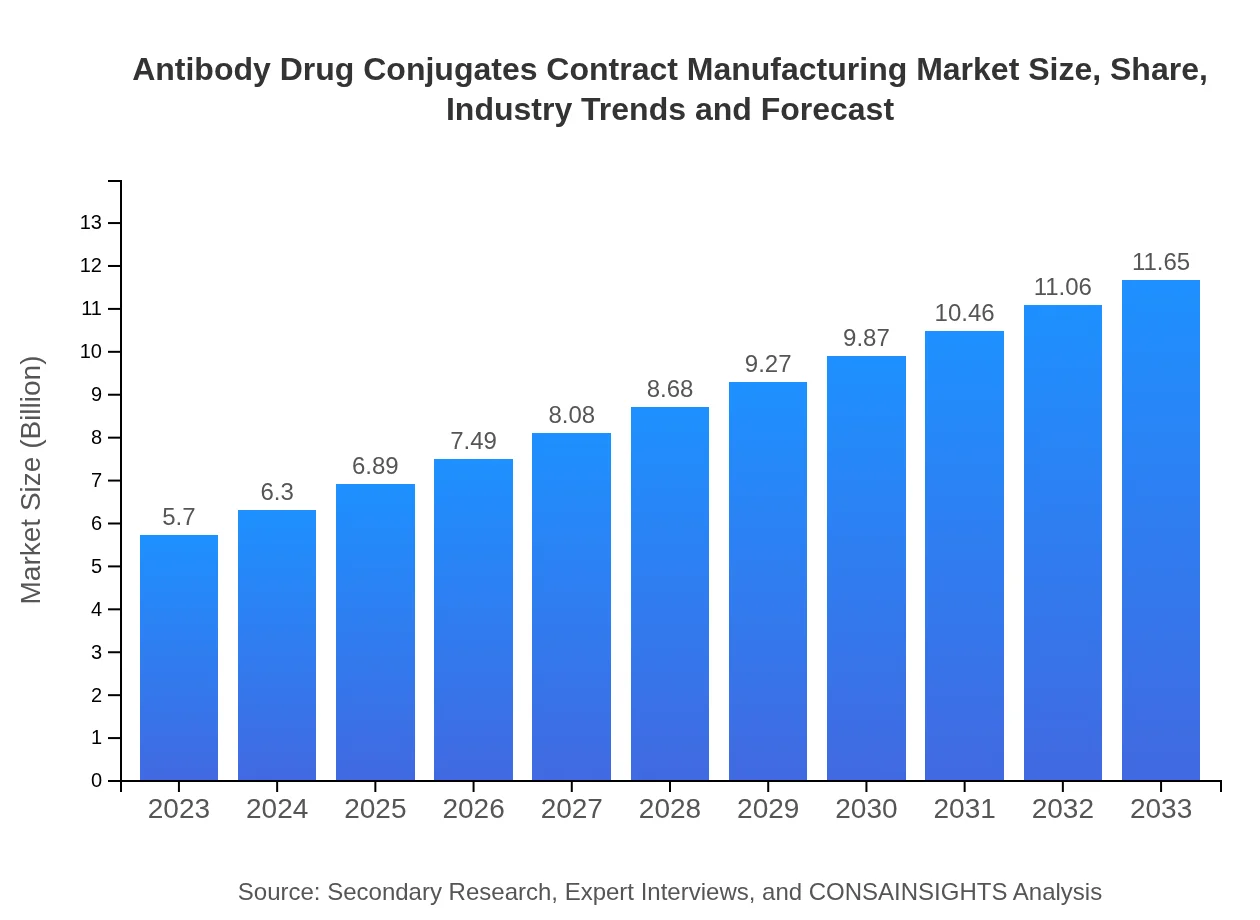
- Market grows from $5.70 Billion in 2023 to $11.65 Billion by 2033, reflecting a 7.2% CAGR through 2023 to 2033.
- North America is the dominant and fastest-expanding region, increasing from $2.01 Billion in 2023 to $4.11 Billion in 2033.
- Europe and Asia Pacific show notable rises: Europe from $1.66 Billion to $3.39 Billion; Asia Pacific from $1.17 Billion to $2.39 Billion.
- Outsourcing demand, regulatory approvals, and technology advances in linker and cytotoxic solutions underpin market expansion; major players include Boehringer Ingelheim, BMS (Bristol-Myers Squibb), and Lonza.
Antibody Drug Conjugates Contract Manufacturing Market Report — Executive Summary
The ADC contract manufacturing sector is poised for sustained expansion, driven by growing demand for targeted therapeutics and more frequent outsourcing by biopharmaceutical developers. The market size rises from $5.70 Billion in 2023 to $11.65 Billion by 2033 at a 7.2% CAGR over 2023 to 2033. Investment in linker and cytotoxic technologies, plus capabilities for small-batch and specialized production, supports capacity growth and competitive differentiation. Regional dynamics favor North America as the largest and fastest-growing market, while Europe and Asia Pacific register steady gains. Service offerings span full-service manufacturing and fill & finish, with regulatory compliance and GMP expertise increasingly decisive. Established contract manufacturers and strategic collaborations are central to meeting rising production complexity and reducing time-to-market for ADC therapies. Leading service providers such as Boehringer Ingelheim, BMS (Bristol-Myers Squibb), and Lonza are key participants shaping capacity and technological investments.
Key Growth Drivers
- Rising demand for targeted oncology therapies is increasing reliance on specialized contract manufacturing capacities.
- Greater outsourcing by biopharmaceutical companies to reduce time-to-market and manage complex ADC production.
- Technological improvements in linker and cytotoxic components enhance manufacturability and therapeutic profiles.
- Regulatory approvals and stricter GMP expectations drive investment in compliant, high-capability contract manufacturing facilities.
- Growth of personalized medicine increases need for adaptable small-batch production and specialized process development.
| Metric | Value |
|---|---|
| Study Period | 2023 - 2033 |
| 2023 Market Size | $5.70 Billion |
| CAGR (2023-2033) | 7.2% |
| 2033 Market Size | $11.65 Billion |
| Top Companies | Boehringer Ingelheim, BMS (Bristol-Myers Squibb), Lonza |
| Last Modified Date | 22 April 2026 |
Antibody Drug Conjugates Contract Manufacturing Market Overview
Customize Antibody Drug Conjugates Contract Manufacturing Market Report market research report
- ✔ Get in-depth analysis of Antibody Drug Conjugates Contract Manufacturing market size, growth, and forecasts.
- ✔ Understand Antibody Drug Conjugates Contract Manufacturing's regional dynamics and industry-specific trends.
- ✔ Identify potential applications, end-user demand, and growth segments in Antibody Drug Conjugates Contract Manufacturing
What is the Market Size & CAGR of Antibody Drug Conjugates Contract Manufacturing Market Report market in 2023?
Antibody Drug Conjugates Contract Manufacturing Industry Analysis
Antibody Drug Conjugates Contract Manufacturing Market Segmentation and Scope
Tell us your focus area and get a customized research report.
Antibody Drug Conjugates Contract Manufacturing Market Report Market Analysis Report by Region
Europe Antibody Drug Conjugates Contract Manufacturing Market Report:
Europe expands from $1.66 Billion in 2023 to $3.39 Billion in 2033. Regional growth is driven by strong oncology research activity, increasing outsourcing to specialized manufacturers, and investments in linker and cytotoxic technology expertise.Asia Pacific Antibody Drug Conjugates Contract Manufacturing Market Report:
Asia Pacific grows from $1.17 Billion in 2023 to $2.39 Billion in 2033. Expansion reflects rising regional clinical development, greater outsourcing uptake by local biopharma firms, and enhanced capacity for specialized ADC production.North America Antibody Drug Conjugates Contract Manufacturing Market Report:
North America is the largest and fastest-growing region, rising from $2.01 Billion in 2023 to $4.11 Billion in 2033. The region benefits from established contract manufacturers, significant adoption of ADC development, and ongoing investments in advanced manufacturing capabilities and compliance.South America Antibody Drug Conjugates Contract Manufacturing Market Report:
Middle East & Africa Antibody Drug Conjugates Contract Manufacturing Market Report:
Middle East and Africa move from $0.35 Billion in 2023 to $0.71 Billion in 2033. Growth stems from incremental demand for specialized therapeutics and the expansion of GMP-capable contract manufacturing services in select markets.Tell us your focus area and get a customized research report.
Research Methodology
Antibody Drug Conjugates Contract Manufacturing Market Analysis By Manufacturing Process
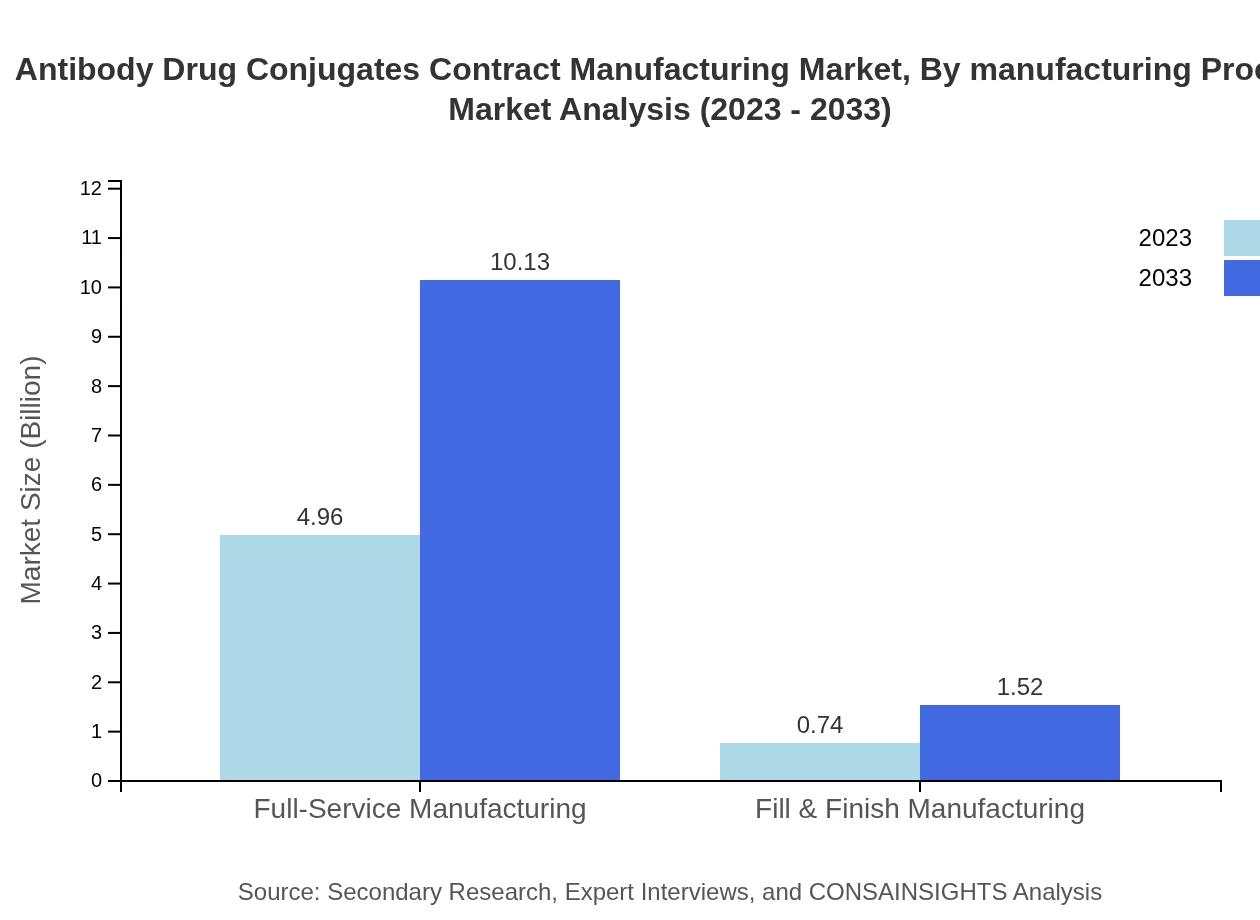
The manufacturing process segment includes full-service manufacturing and fill & finish services, with full-service manufacturing nearing $4.96 billion in 2023 and estimated to grow to $10.13 billion by 2033, capturing 86.95% share due to the preference for comprehensive service offerings by clients. Conversely, fill & finish manufacturing, while vital, holds a smaller market share, growing cautiously from $0.74 billion to $1.52 billion in the same period.
Antibody Drug Conjugates Contract Manufacturing Market Analysis By Therapeutic Area
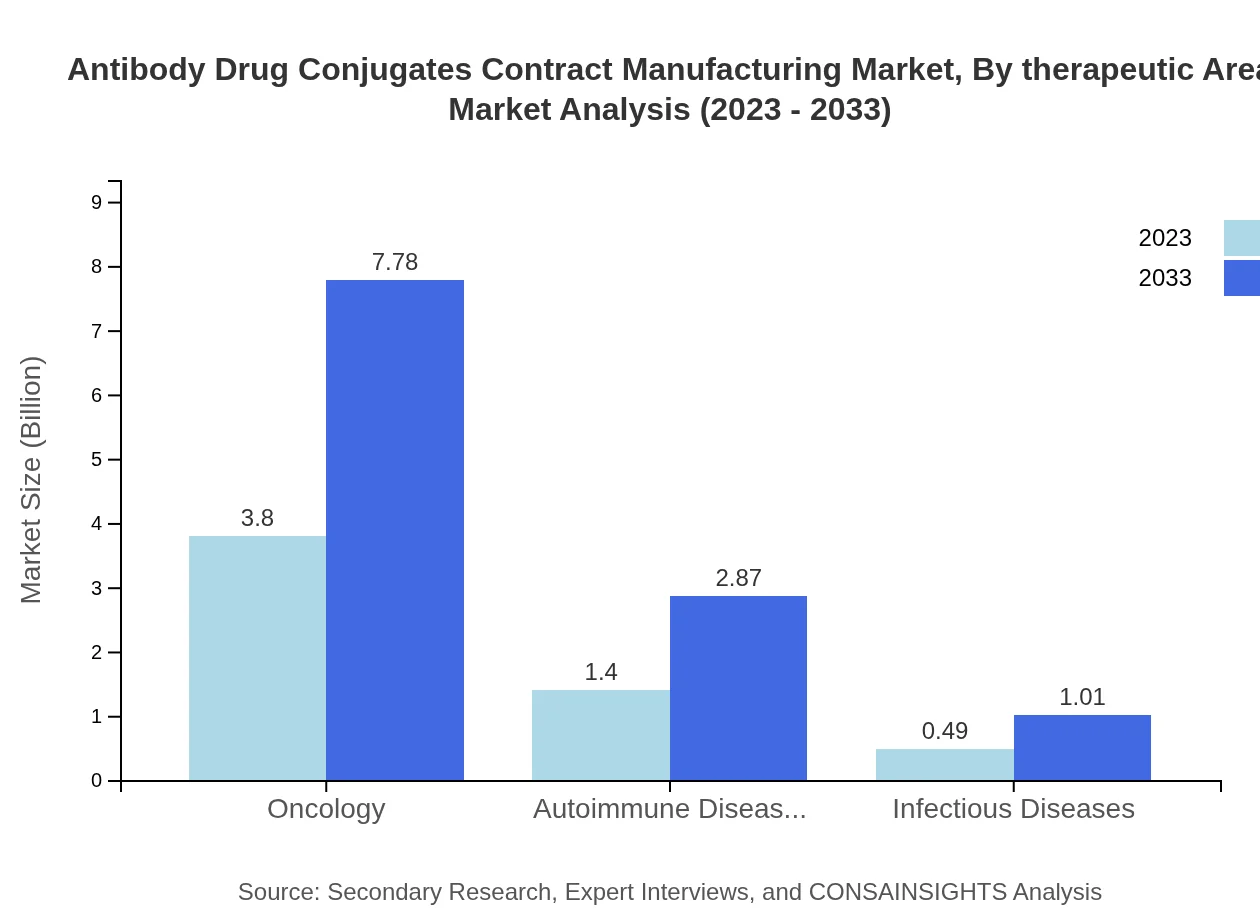
Focusing on therapeutic areas, oncology dominates with a significant market size increase from $3.80 billion in 2023 to $7.78 billion by 2033, maintaining a 66.75% share. Autoimmune diseases follow with growing segments, increasing from $1.40 billion to $2.87 billion. Infectious diseases represent a smaller segment, projected to rise from $0.49 billion to $1.01 billion.
Antibody Drug Conjugates Contract Manufacturing Market Analysis By Technology
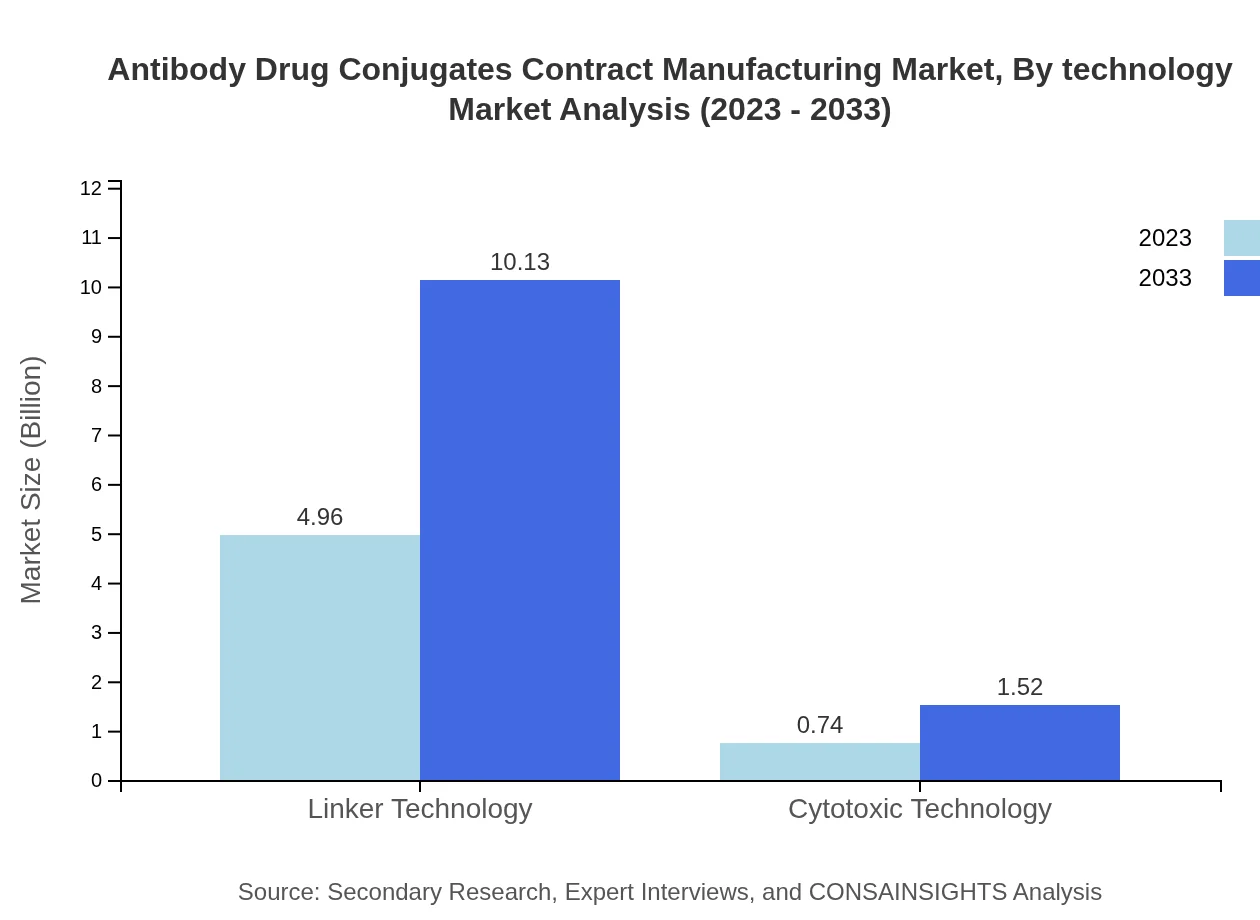
Linker technology and cytotoxic technology lead the technological segment, with linker technology growth mirroring the overall market from $4.96 billion to $10.13 billion. Cytotoxic technology demonstrates slower growth, from $0.74 billion to $1.52 billion, yet holds significant strategic relevance in developing high-efficacy ADCs.
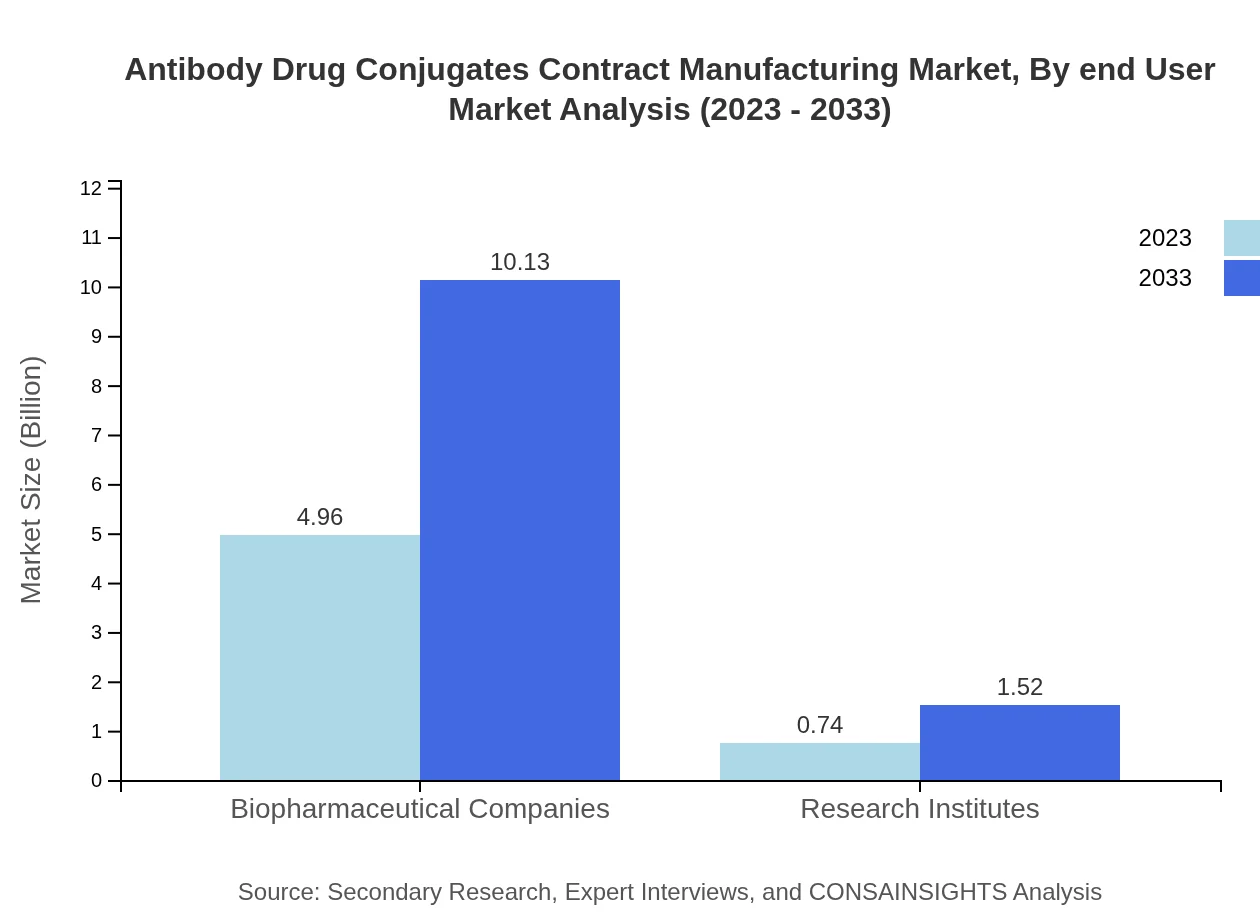
Antibody Drug Conjugates Contract Manufacturing Market Analysis By End User
The end-user segment showcases strong demand from biopharmaceutical companies, expected to continue its dominance, with growth from $4.96 billion to $10.13 billion. Research institutes also contribute significantly, with a market increase from $0.74 billion to $1.52 billion, reflecting a collaborative effort in drug development.
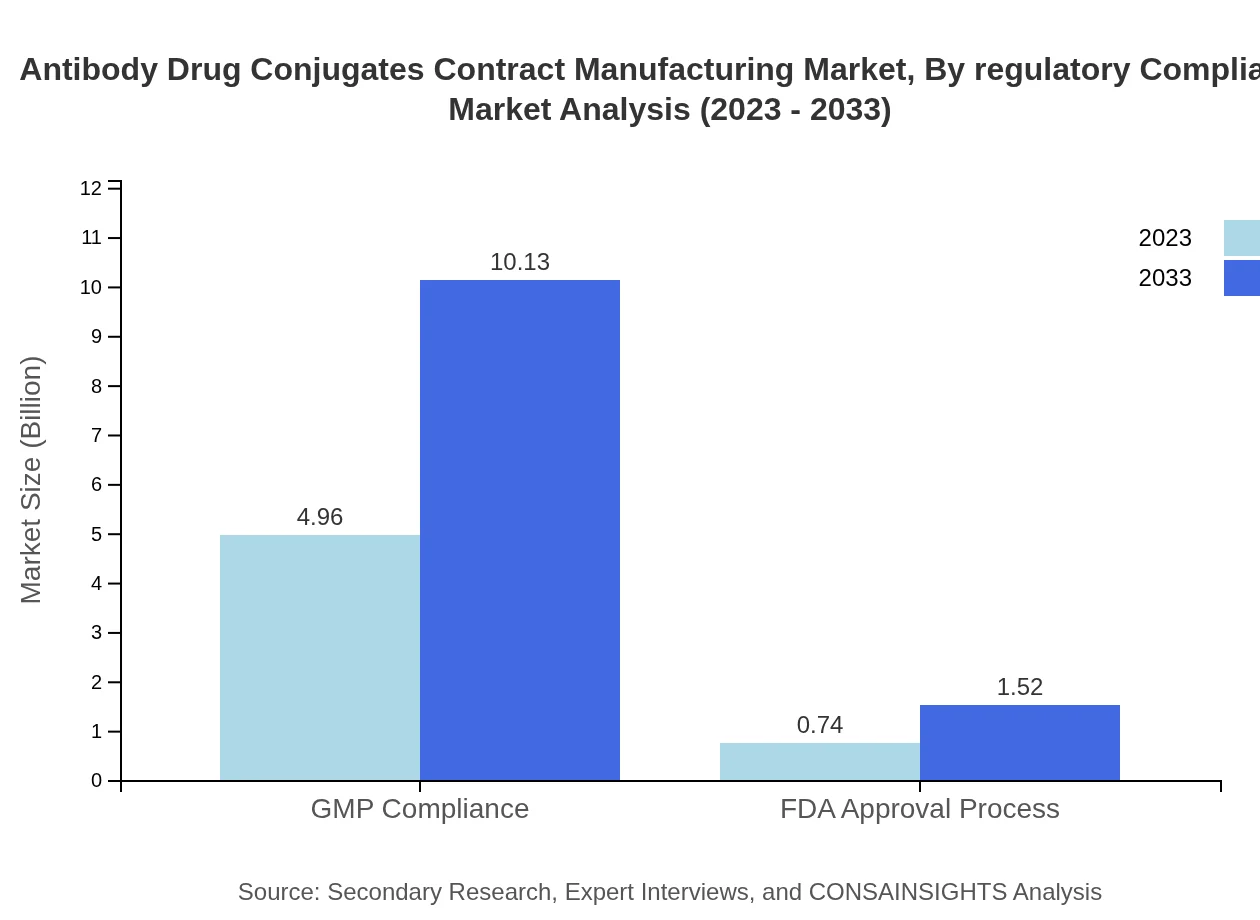
Antibody Drug Conjugates Contract Manufacturing Market Analysis By Regulatory Compliance
Regulatory compliance remains a critical factor, with GMP compliance ensuring safety and efficacy standards representing a giant segment at $4.96 billion growing to $10.13 billion. FDA approval processes follow, growing from $0.74 billion to $1.52 billion, emphasizing the importance of regulatory adherence in ADC manufacture.
Antibody Drug Conjugates Contract Manufacturing Market Trends and Future Forecast
Tell us your focus area and get a customized research report.
Global Market Leaders and Top Companies in Antibody Drug Conjugates Contract Manufacturing Industry
Boehringer Ingelheim:
Boehringer Ingelheim is a leading global player in biopharmaceutical contract manufacturing, offering innovative solutions for ADCs focusing on efficiency and quality control throughout the manufacturing cycle.BMS (Bristol-Myers Squibb):
Bristol-Myers Squibb has invested extensively in therapeutic development and utilizes ADCs as part of their cancer treatment portfolio, driving demand for contract manufacturing services that ensure high-quality production standards.Lonza:
Lonza is recognized for its integrated offerings in contract manufacturing, specializing in complex biopharmaceuticals including ADCs, with state-of-the-art facilities that enhance production capabilities.We're grateful to work with incredible clients.









FAQs
What is the market size of the ADC contract manufacturing market in 2023?
The market size in 2023 is $5.70 Billion. This figure reflects current contract manufacturing demand for ADCs and serves as the baseline for projections through 2033.
How big will the market be by 2033?
By 2033 the market is projected to reach $11.65 Billion, based on the forecast covering 2023 to 2033 and reflecting ongoing adoption of ADC therapies.
What is CAGR for the forecast period?
The compound annual growth rate for the forecast period 2023 to 2033 is 7.2%, representing steady expansion driven by outsourcing and technological advances.
Why is North America important in this market?
North America is both the largest and fastest-growing region, supported by established contract manufacturers, investment in advanced capabilities, and significant ADC development activity.
Which regions show notable growth in the forecast?
Europe and Asia Pacific show meaningful increases: Europe grows from $1.66 Billion in 2023 to $3.39 Billion in 2033; Asia Pacific rises from $1.17 Billion to $2.39 Billion.
Who are some leading contract manufacturing companies in this space?
Top companies active in ADC contract manufacturing include Boehringer Ingelheim, BMS (Bristol-Myers Squibb), and Lonza, which invest in specialized capabilities and regulatory compliance.
What manufacturing services are commonly offered?
Common service models include full-service manufacturing and fill & finish manufacturing, addressing end-to-end process needs and late-stage product finishing for ADC developers.
Why are linker and cytotoxic technologies significant?
Advances in linker and cytotoxic technologies improve ADC stability, targeting, and manufacturability, influencing production complexity and the services required from contract manufacturers.
What regulatory capabilities are prioritized by manufacturers?
GMP compliance and navigation of the FDA approval process are prioritized, ensuring quality, meeting regulatory standards, and facilitating clinical and commercial supply.
How does personalized medicine affect contract manufacturing demand?
Personalized medicine boosts demand for tailored, small-batch production and flexible manufacturing processes, prompting investment in adaptable technologies and specialized workflows.