Biocompatibility Testing Services Market Report
Published Date: 22 April 2026 | Report Code: biocompatibility-testing-services
Biocompatibility Testing Services Market Size, Share, Industry Trends and Forecast to 2033
This report provides an in-depth analysis of the Biocompatibility Testing Services market, covering market size, growth forecasts, regional insights, segmentation analysis, and trends from 2023 to 2033.
Key Takeaways
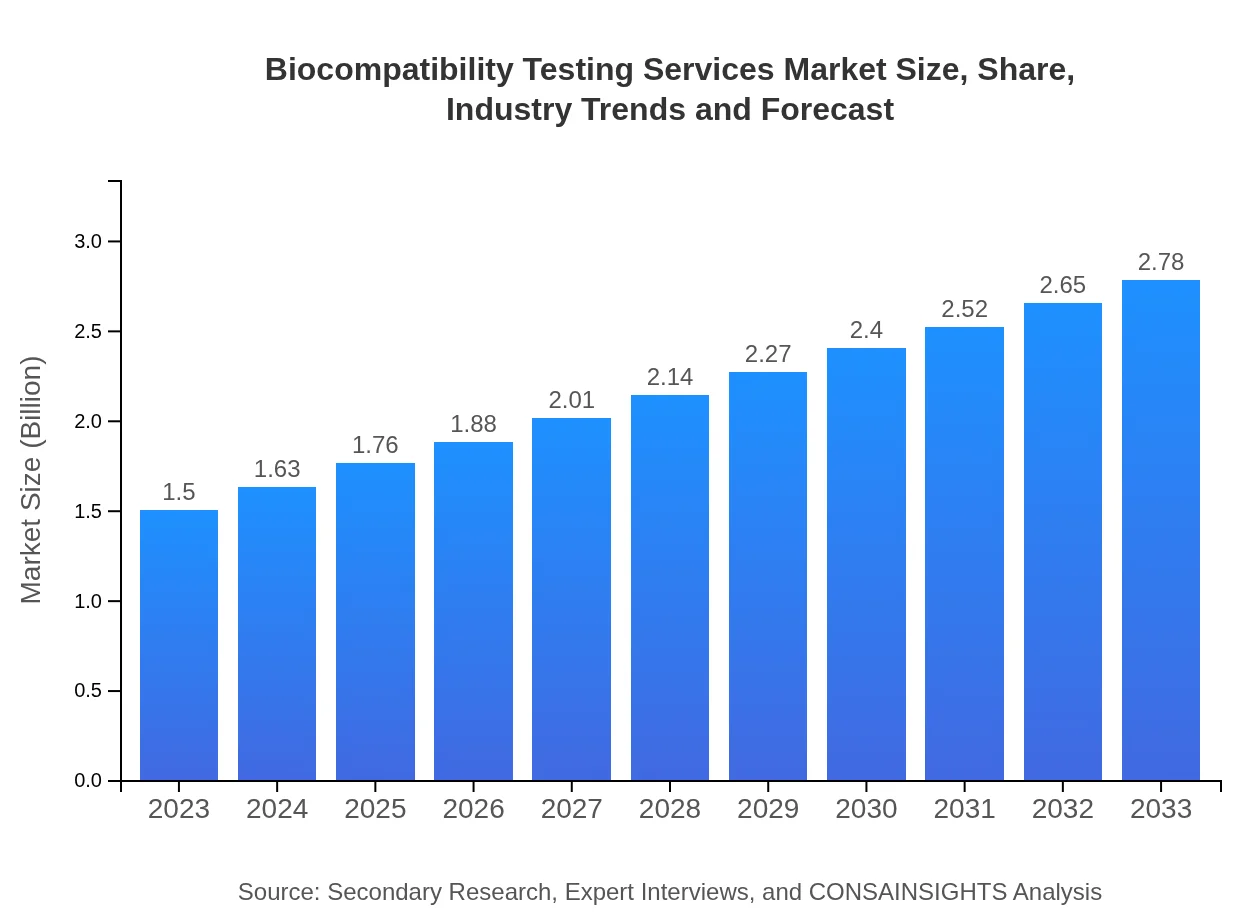
- Global market was $1.50 Billion in 2023 and is forecast to reach $2.78 Billion by 2033 at a 6.2% CAGR.
- North America is both the largest and the fastest-growing region, with demand rising from $0.54 Billion to $1 Billion.
- Europe and Asia Pacific show steady expansion, with Europe moving from $0.46 Billion to $0.86 Billion and Asia Pacific from $0.28 Billion to $0.51 Billion.
- Leading providers include Eurofins Scientific, Charles River Laboratories, NAMSA and TÜV Rheinland, shaping capacity and service offerings.
Biocompatibility Testing Services Market Report — Executive Summary
The market for biocompatibility testing services is expanding as regulatory scrutiny and safety expectations increase across medical device and pharmaceutical sectors. The market measured $1.50 Billion in 2023 and is projected to grow to $2.78 Billion by 2033 at a 6.2% CAGR for the 2023 to 2033 period. Drivers include stricter compliance requirements, growing R&D investment, and adoption of advanced testing approaches such as in vitro, in vivo and computational modeling. North America leads in scale and pace, supported by dense industry activity and testing infrastructure. Key providers—Eurofins Scientific, Charles River Laboratories, NAMSA and TÜV Rheinland—are prominent in delivering services to manufacturers and CROs. Competitive dynamics emphasize service specialization, validation capabilities and regulatory expertise, while sustainability and methodological innovations shape longer-term market evolution.
Key Growth Drivers
- Increasing regulatory stringency for medical devices and pharmaceuticals driving demand for validated testing.
- Rising R&D expenditure in life sciences expands the need for preclinical and compatibility assessments.
- Advances in testing methodologies, including computational and in vitro techniques, improve throughput and accuracy.
- Consolidation and capacity-building by major providers enhance service availability and quality across regions.
| Metric | Value |
|---|---|
| Study Period | 2023 - 2033 |
| 2023 Market Size | $1.50 Billion |
| CAGR (2023-2033) | 6.2% |
| 2033 Market Size | $2.78 Billion |
| Top Companies | Eurofins Scientific, Charles River Laboratories, NAMSA, TÜV Rheinland |
| Last Modified Date | 22 April 2026 |
Biocompatibility Testing Services Market Overview
Customize Biocompatibility Testing Services Market Report market research report
- ✔ Get in-depth analysis of Biocompatibility Testing Services market size, growth, and forecasts.
- ✔ Understand Biocompatibility Testing Services's regional dynamics and industry-specific trends.
- ✔ Identify potential applications, end-user demand, and growth segments in Biocompatibility Testing Services
What is the Market Size & CAGR of Biocompatibility Testing Services Market Report market in 2023?
Biocompatibility Testing Services Industry Analysis
Biocompatibility Testing Services Market Segmentation and Scope
Tell us your focus area and get a customized research report.
Biocompatibility Testing Services Market Report Market Analysis Report by Region
Europe Biocompatibility Testing Services Market Report:
Europe grows from $0.46 Billion in 2023 to $0.86 Billion in 2033. Regional expansion is supported by regulatory compliance requirements, a strong presence of manufacturers and CROs, and investments in validated testing protocols.Asia Pacific Biocompatibility Testing Services Market Report:
Asia Pacific increases from $0.28 Billion in 2023 to $0.51 Billion in 2033. Growth is driven by rising healthcare investment, expanding manufacturing activity and growing adoption of standardized testing methods across markets.North America Biocompatibility Testing Services Market Report:
North America is both the largest and fastest-growing region, expanding from $0.54 Billion in 2023 to $1 Billion in 2033. The region benefits from concentrated medical device and pharmaceutical R&D, stringent regulatory oversight and established testing infrastructure that sustain demand.South America Biocompatibility Testing Services Market Report:
Middle East & Africa Biocompatibility Testing Services Market Report:
Middle East and Africa expands from $0.09 Billion in 2023 to $0.16 Billion in 2033. Market progress reflects growing healthcare investment, regulatory harmonization efforts and increasing use of third-party testing services.Tell us your focus area and get a customized research report.
Research Methodology
Biocompatibility Testing Services Market Analysis By Service Type
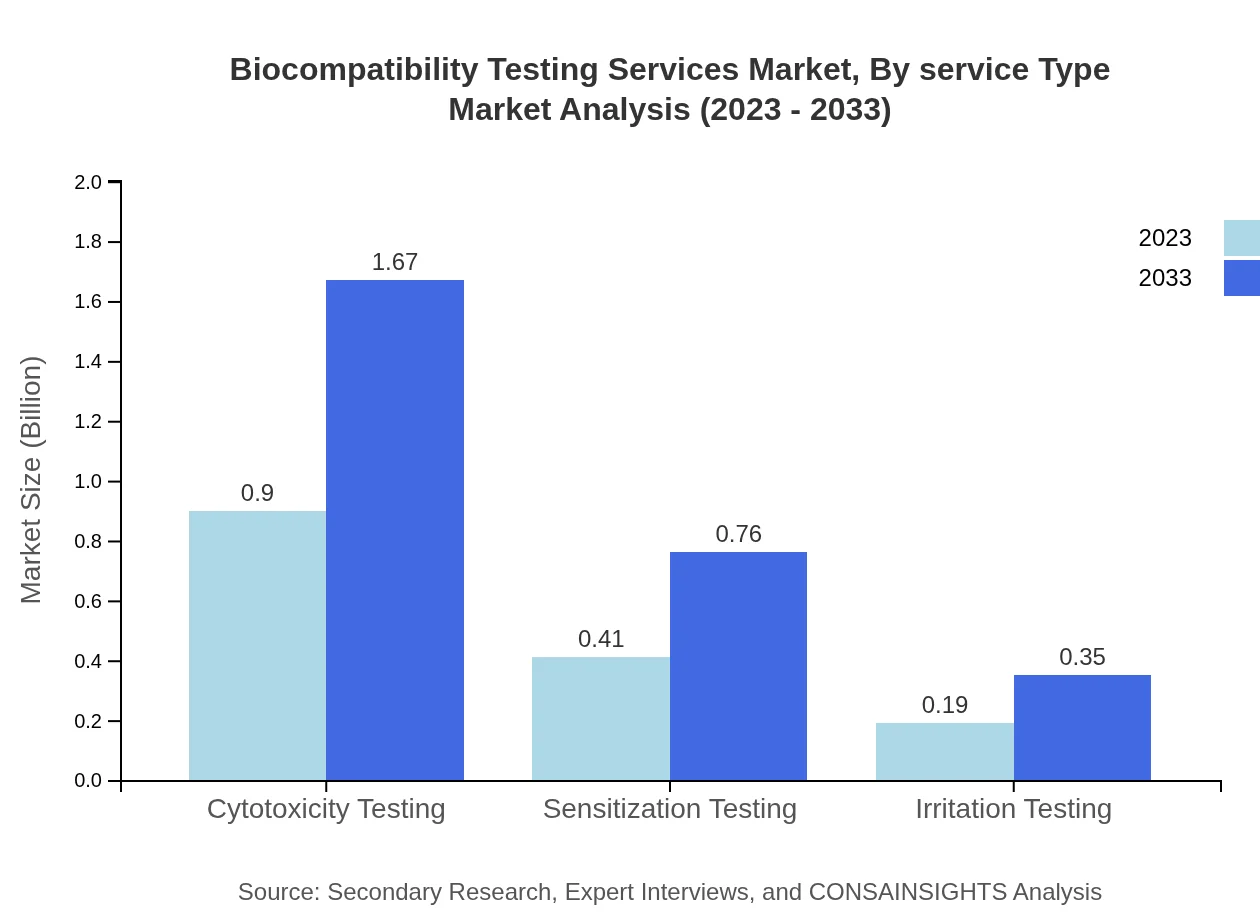
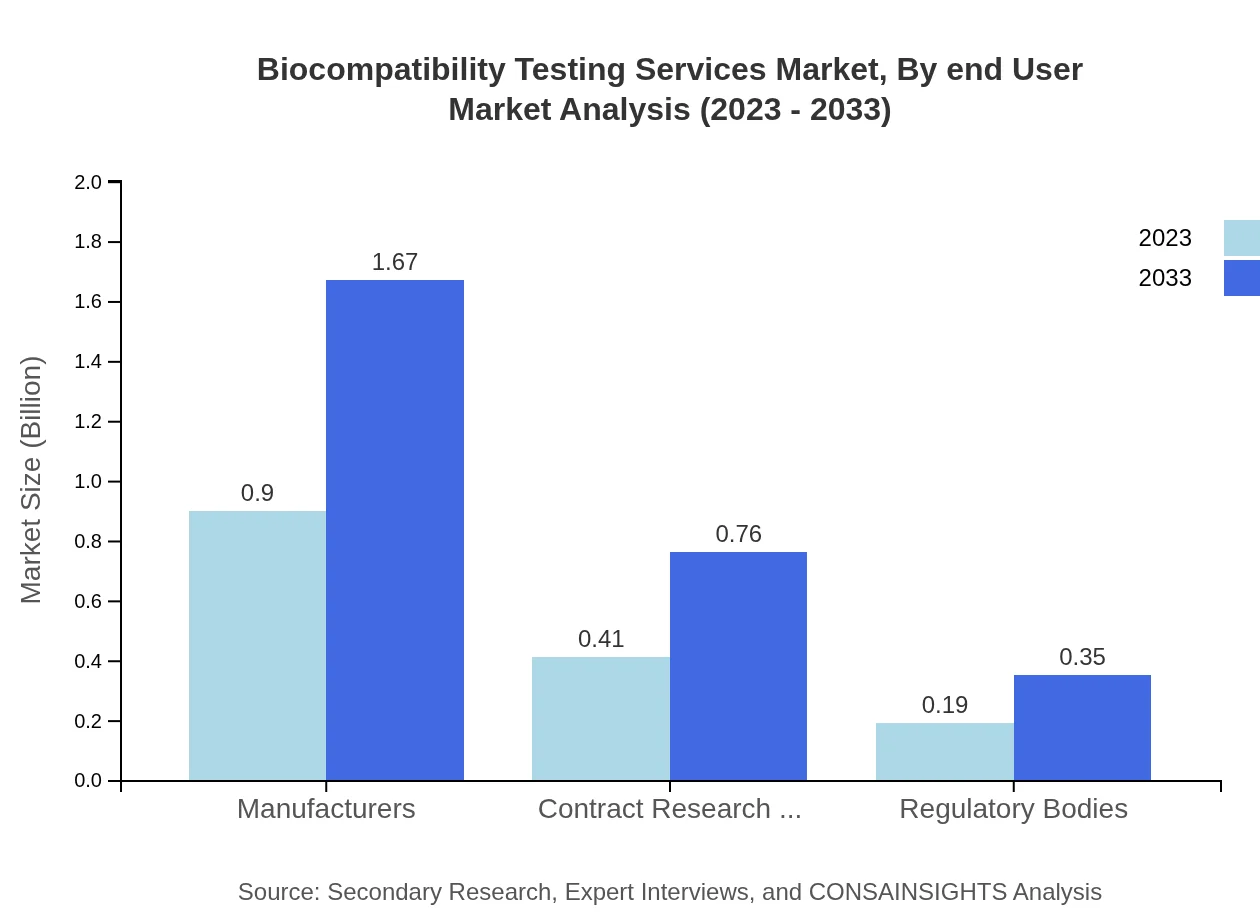
The Biocompatibility Testing Services Market, segmented by service type, highlights that manufacturers hold the largest market share, expected to grow from $0.90 billion in 2023 to $1.67 billion by 2033, capturing 60.2% of the market. CROs follow behind, with an increasing market size from $0.41 billion to $0.76 billion, thereby maintaining 27.35% market share. Regulatory bodies account for $0.19 billion, growing to $0.35 billion, capturing 12.45%. This segmentation showcases the reliance on established manufacturers and CROs for accurate testing services.
Biocompatibility Testing Services Market Analysis By Industry
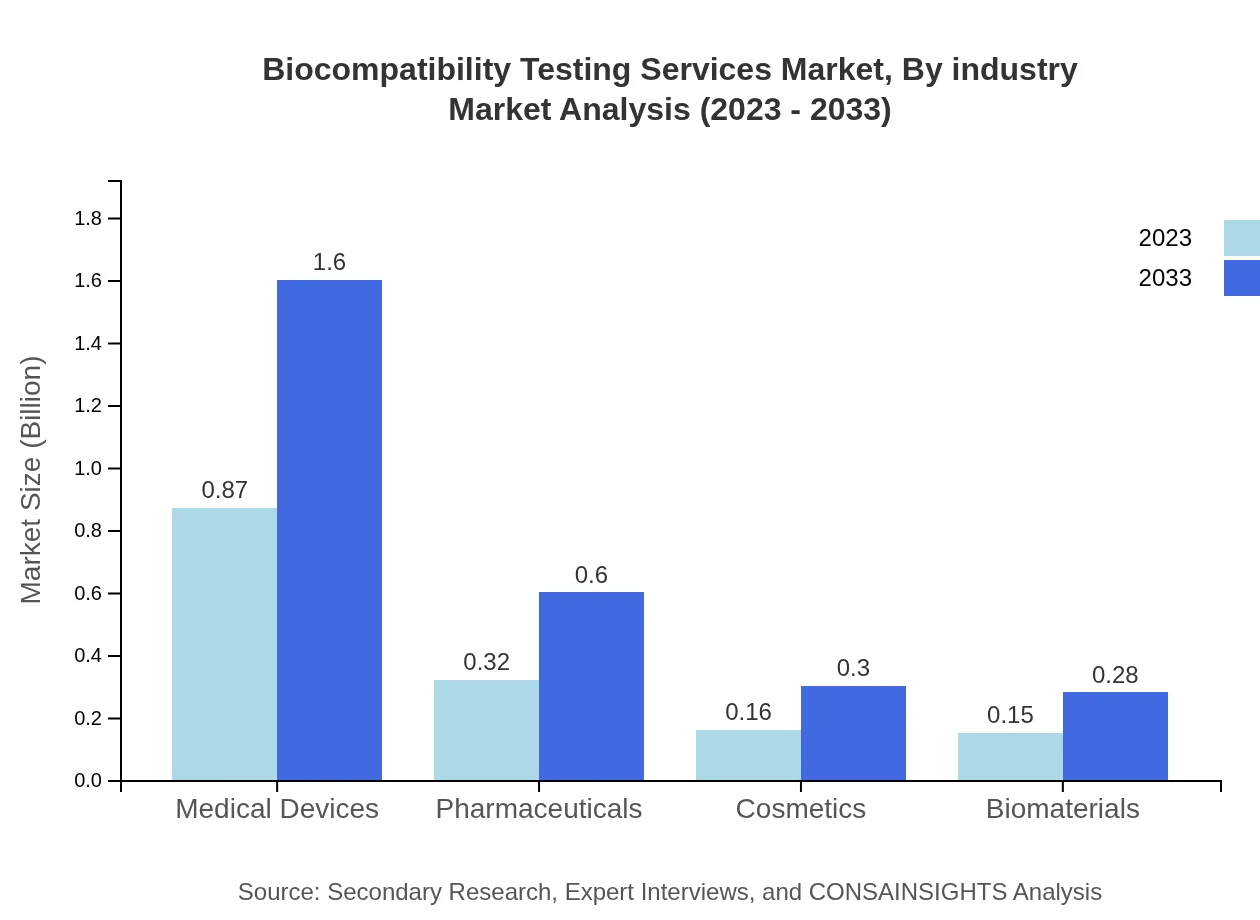
In the Biocompatibility Testing Services Market, industry segmentation indicates that medical devices dominate with a market size rising from $0.87 billion in 2023 to $1.60 billion by 2033, accounting for a 57.69% share. Pharmaceuticals will see growth from $0.32 billion to $0.60 billion for a 21.43% share, while cosmetics contribute $0.16 billion to $0.30 billion and biomaterials progress from $0.15 billion to $0.28 billion, each capturing around 10% shares.
Biocompatibility Testing Services Market Analysis By Test Methodology
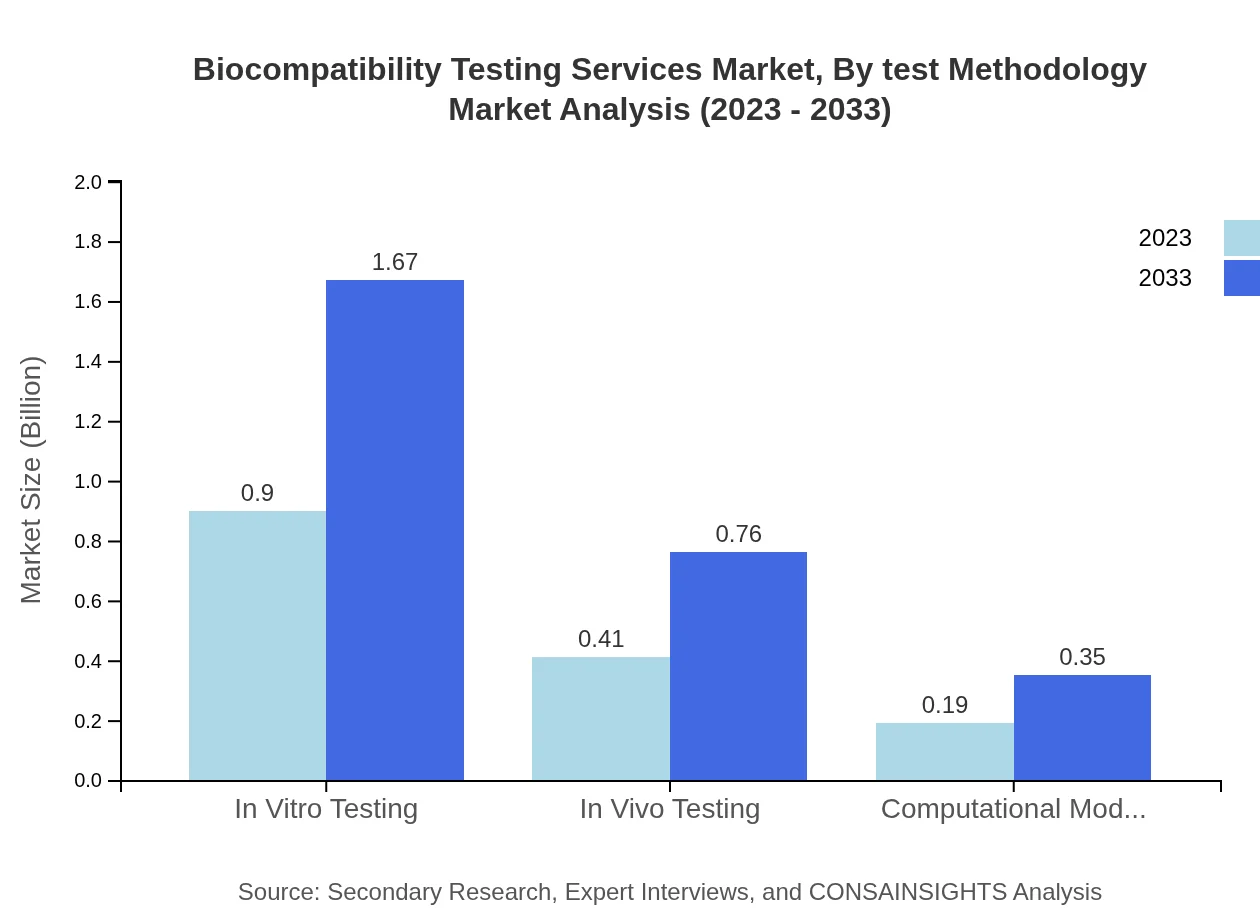
The testing methodology segment reveals that In Vitro Testing leads with a sizable market share, estimated at $0.90 billion growing to $1.67 billion, and accounting for 60.2%. In Vivo Testing grows from $0.41 billion to $0.76 billion, representing 27.35%. Computational Modeling also maintains a steady growth from $0.19 billion to $0.35 billion, capturing 12.45% market share, indicating a trend towards more technologically advanced testing methodologies.
Biocompatibility Testing Services Market Analysis By End User
The end-user analysis shows that major players in the medical device sector leverage biocompatibility testing extensively, with a market size of $2.00 billion in 2023 projected to reach $3.50 billion in 2033. Pharmaceuticals will extensively utilize these services, with the market expanding from $0.40 billion to $0.80 billion. The cosmetics sector is expected to see utilization increasing from $0.20 billion to $0.30 billion, showcasing the diverse applications of biocompatibility testing across sectors.
Biocompatibility Testing Services Market Trends and Future Forecast
Tell us your focus area and get a customized research report.
Global Market Leaders and Top Companies in Biocompatibility Testing Services Industry
Eurofins Scientific:
Eurofins Scientific is a global group of laboratories known for its commitment to quality and expertise in biocompatibility testing and analytical services, catering to the pharmaceutical and medical device sectors.Charles River Laboratories:
Charles River is a prominent provider of early-stage contract research services, offering comprehensive biocompatibility testing to support their clients in accelerating the development and approval of new pharmaceutical and biotechnology products.NAMSA:
Named as a leader in biocompatibility testing, NAMSA specializes in preclinical and clinical research services, helping manufacturers bring safe and effective medical devices to market.TÜV Rheinland:
TÜV Rheinland is a major player in the field, providing testing and certification to ensure products meet global standards for quality and safety, including those in biocompatibility.We're grateful to work with incredible clients.









FAQs
What is the market size in 2023?
The market size for biocompatibility testing services in 2023 is $1.50 Billion, reflecting demand from medical device and pharmaceutical sectors for safety assessments and regulatory compliance as reported in the data.
How big will the market be in 2033?
The market is projected to reach $2.78 Billion by 2033, reflecting steady expansion over the 2023 to 2033 forecast period driven by regulatory requirements and testing technology improvements.
What is CAGR of the Biocompatibility Testing Services Market Report?
The reported compound annual growth rate is 6.2% for the forecast period 2023 to 2033, indicating moderate, sustained growth driven by compliance demands, R&D spending and methodological innovation.
Why is North America the largest region?
North America is the largest and fastest-growing market due to concentrated medical device and pharmaceutical activity, regulatory rigor and high testing demand; it grows from $0.54 Billion in 2023 to $1 Billion in 2033.
Which regions are showing fastest growth?
North America is identified as the fastest-growing region, increasing from $0.54 Billion in 2023 to $1 Billion in 2033; other regions also report growth across the 2023 to 2033 period.
Who are the leading companies in this market?
Top companies named in the input are Eurofins Scientific, Charles River Laboratories, NAMSA and TÜV Rheinland, which are notable service providers across testing, validation and regulatory support functions.
What are common test methodologies used?
Common methodologies cited include In Vitro Testing, In Vivo Testing and Computational Modeling, representing laboratory experiments and modeling approaches used to evaluate material and device compatibility.
What service types are offered in this market?
Service types specified include Cytotoxicity Testing, Sensitization Testing and Irritation Testing, covering core assessments required for demonstrating biocompatibility under regulatory frameworks.