Biopharmaceuticals Contract Manufacturing Market Report
Published Date: 22 April 2026 | Report Code: biopharmaceuticals-contract-manufacturing
Biopharmaceuticals Contract Manufacturing Market Size, Share, Industry Trends and Forecast to 2033
This report provides an in-depth analysis of the Biopharmaceuticals Contract Manufacturing market, offering insights into market trends, segmentation, regional analysis, and key players from 2023 to 2033.
Key Takeaways
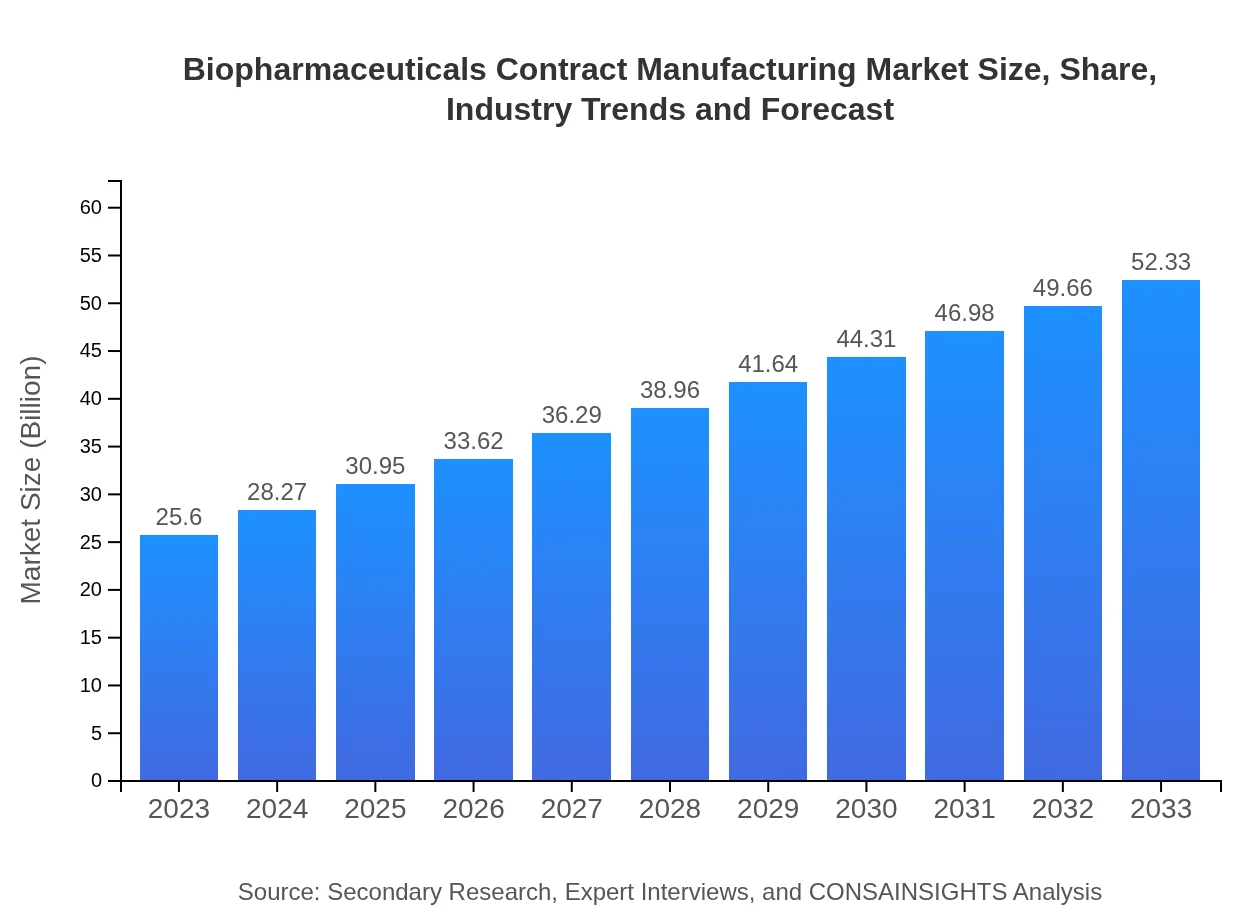
- Global market expands from $25.60 Billion in 2023 to $52.33 Billion by 2033 at a 7.2% CAGR, reflecting sustained demand for outsourced biologics manufacturing.
- North America is both the largest and fastest-growing region, with its scale and investment driving capacity and service breadth.
- Europe and Asia Pacific show notable growth: Europe from $7.81 Billion to $15.97 Billion, Asia Pacific from $4.82 Billion to $9.84 Billion.
- Leading contract manufacturers such as Lonza Group, Samsung Biologics, and WuXi AppTec are central to capacity expansion and specialized services.
- Regulatory frameworks and compliance requirements (FDA, EMEA, CGMP) are shaping service offerings and quality systems across regions.
Biopharmaceuticals Contract Manufacturing Market Report — Executive Summary
The Biopharmaceuticals Contract Manufacturing Market Report details steady sector expansion driven by rising biologics development and strategic outsourcing. The market rises from $25.60 Billion in 2023 to $52.33 Billion by 2033 at a 7.2% CAGR, with North America leading in size and growth. Demand for specialized services—upstream cell culture, purification, and formulation and filling—supports CDMO investment. Key product focuses include monoclonal antibodies, recombinant proteins, vaccines, gene therapy products, and other biologics. End users span biopharmaceutical companies, academic research organizations, and government bodies, while regulatory compliance (CGMP, FDA, EMEA) remains a priority. Major providers such as Lonza Group, Samsung Biologics, and WuXi AppTec are expanding capabilities to meet complex biologics requirements. The report examines segment-level dynamics, regional trajectories, and competitive positioning to inform strategic decisions by stakeholders.
Key Growth Drivers
- Rising biologics development prompts biopharmaceutical firms to outsource complex manufacturing processes to specialized CDMOs.
- Strategic focus on core R&D leads companies to contract manufacturing to accelerate timelines and control capital expenditure.
- Technological advances in cell culture, purification, and formulation enable higher-yield, scalable production for diverse biologic modalities.
- Stringent regulatory standards (CGMP, FDA, EMEA) increase demand for experienced contract manufacturers with validated quality systems.
- Regional investment, especially in North America, is expanding capacity and specialized service offerings to support global supply needs.
| Metric | Value |
|---|---|
| Study Period | 2023 - 2033 |
| 2023 Market Size | $25.60 Billion |
| CAGR (2023-2033) | 7.2% |
| 2033 Market Size | $52.33 Billion |
| Top Companies | Lonza Group, Samsung Biologics, WuXi AppTec |
| Last Modified Date | 22 April 2026 |
Biopharmaceuticals Contract Manufacturing Market Overview
Customize Biopharmaceuticals Contract Manufacturing Market Report market research report
- ✔ Get in-depth analysis of Biopharmaceuticals Contract Manufacturing market size, growth, and forecasts.
- ✔ Understand Biopharmaceuticals Contract Manufacturing's regional dynamics and industry-specific trends.
- ✔ Identify potential applications, end-user demand, and growth segments in Biopharmaceuticals Contract Manufacturing
What is the Market Size & CAGR of Biopharmaceuticals Contract Manufacturing Market Report market in 2023?
Biopharmaceuticals Contract Manufacturing Industry Analysis
Biopharmaceuticals Contract Manufacturing Market Segmentation and Scope
Tell us your focus area and get a customized research report.
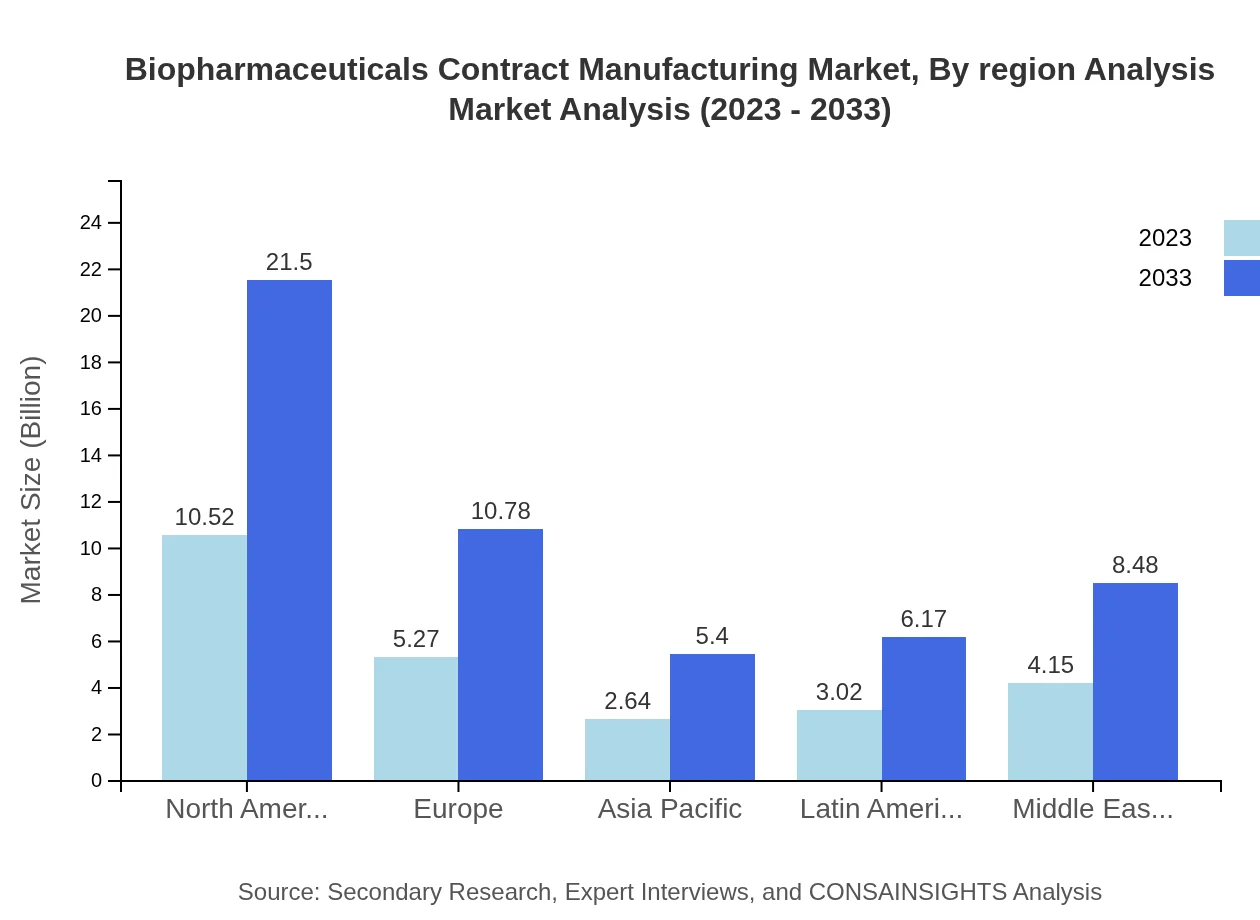
Biopharmaceuticals Contract Manufacturing Market Report Market Analysis Report by Region
Europe Biopharmaceuticals Contract Manufacturing Market Report:
Europe grows from $7.81 Billion in 2023 to $15.97 Billion in 2033. A mature regulatory environment and established manufacturing bases drive demand for contract services in monoclonal antibodies, recombinant proteins, and vaccine production.Asia Pacific Biopharmaceuticals Contract Manufacturing Market Report:
Asia Pacific expands from $4.82 Billion in 2023 to $9.84 Billion in 2033. Rising biopharma activity, capacity additions, and increased outsourcing to CDMOs are key regional growth drivers supporting diverse biologics manufacturing.North America Biopharmaceuticals Contract Manufacturing Market Report:
North America is the largest and fastest-growing region, increasing from $9.33 Billion in 2023 to $19.08 Billion in 2033. Strong industry presence, investment in specialized facilities, and client demand for advanced biologics manufacturing underpin regional expansion.South America Biopharmaceuticals Contract Manufacturing Market Report:
Middle East & Africa Biopharmaceuticals Contract Manufacturing Market Report:
Middle East and Africa advance from $2.48 Billion in 2023 to $5.08 Billion in 2033. Investment in infrastructure, focus on supply security, and partnerships with established CDMOs are supporting market development across these regions.Tell us your focus area and get a customized research report.
Research Methodology
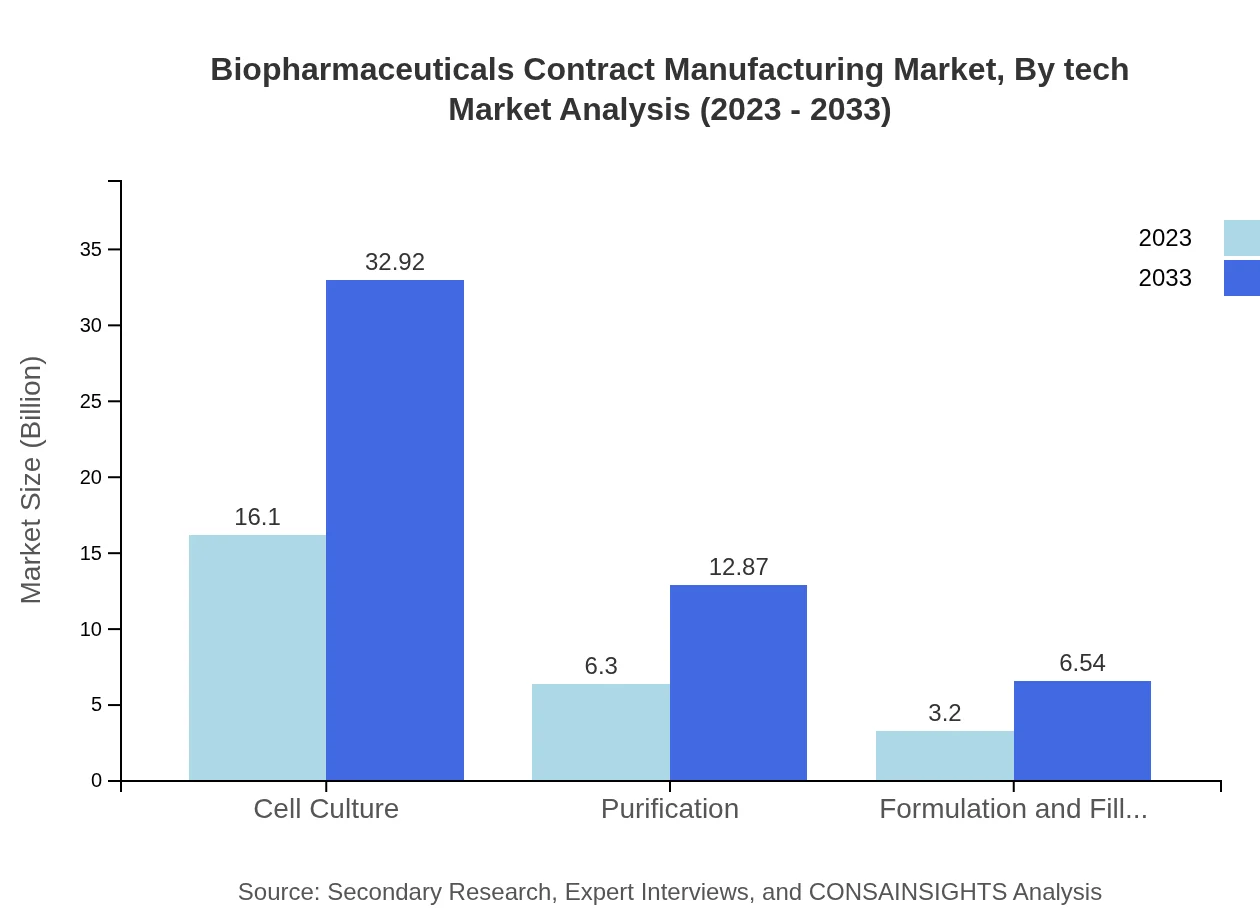
Biopharmaceuticals Contract Manufacturing Market Analysis By Tech
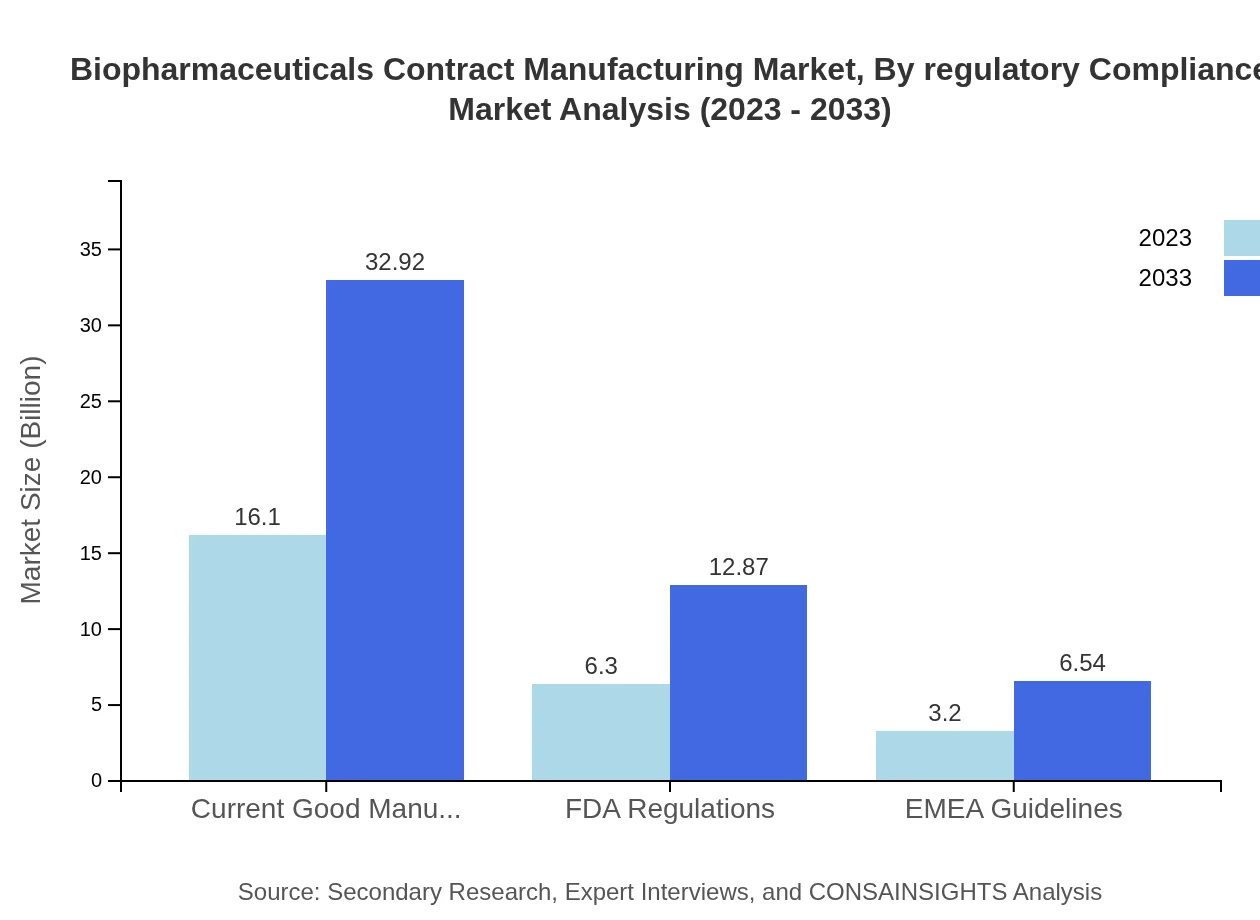
The key technology segments shaping the biopharmaceuticals contract manufacturing market include cell culture and purification processes. Cell culture technology dominates the market due to its extensive application in producing mammalian cell lines for biopharmaceuticals, with a market size projected to grow from $16.10 billion in 2023 to $32.92 billion by 2033.
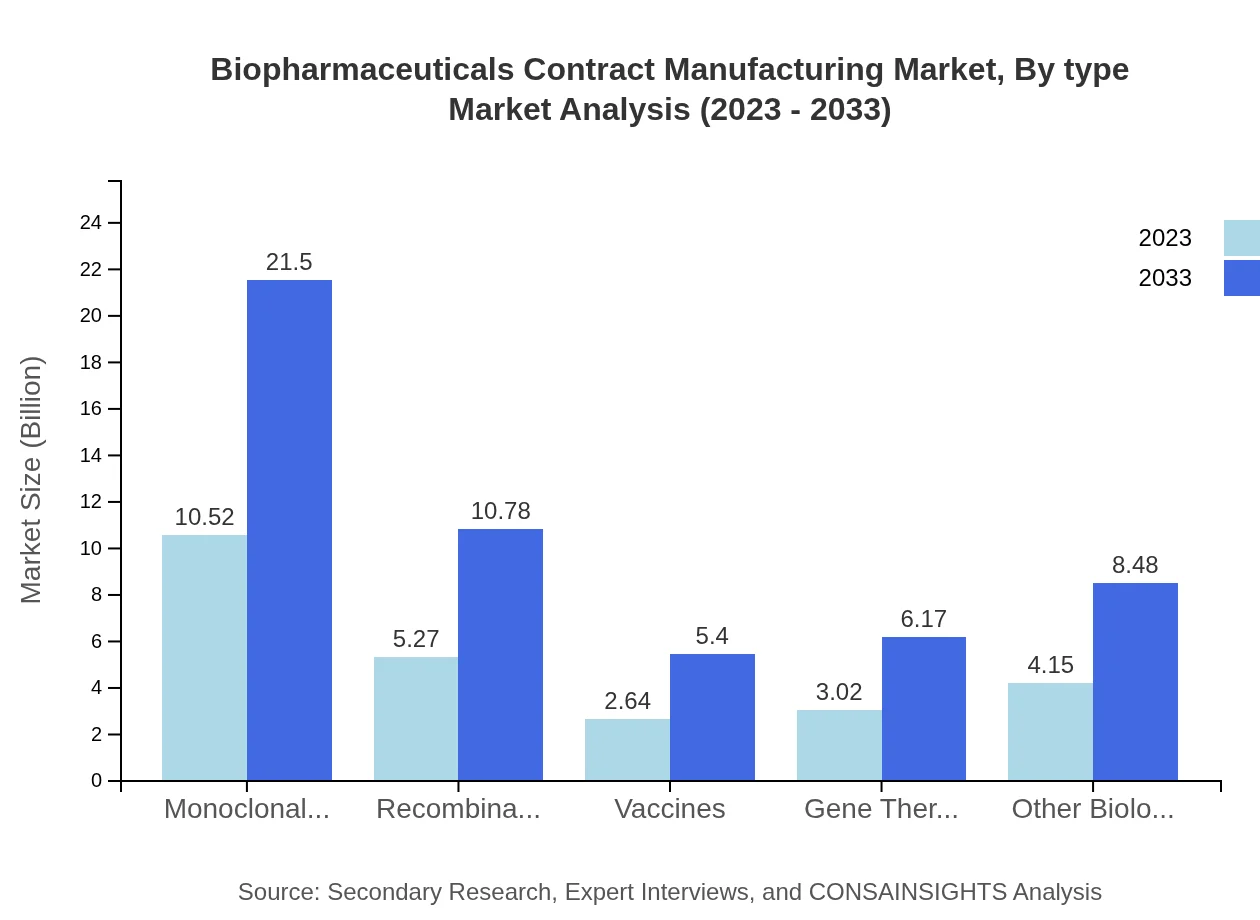
Biopharmaceuticals Contract Manufacturing Market Analysis By Type
Product types such as monoclonal antibodies, recombinant proteins, and vaccines are crucial in the biopharmaceuticals contract manufacturing landscape. Monoclonal antibodies' market size is expected to reach $21.50 billion by 2033 from $10.52 billion in 2023, highlighting their significance in therapeutic applications.
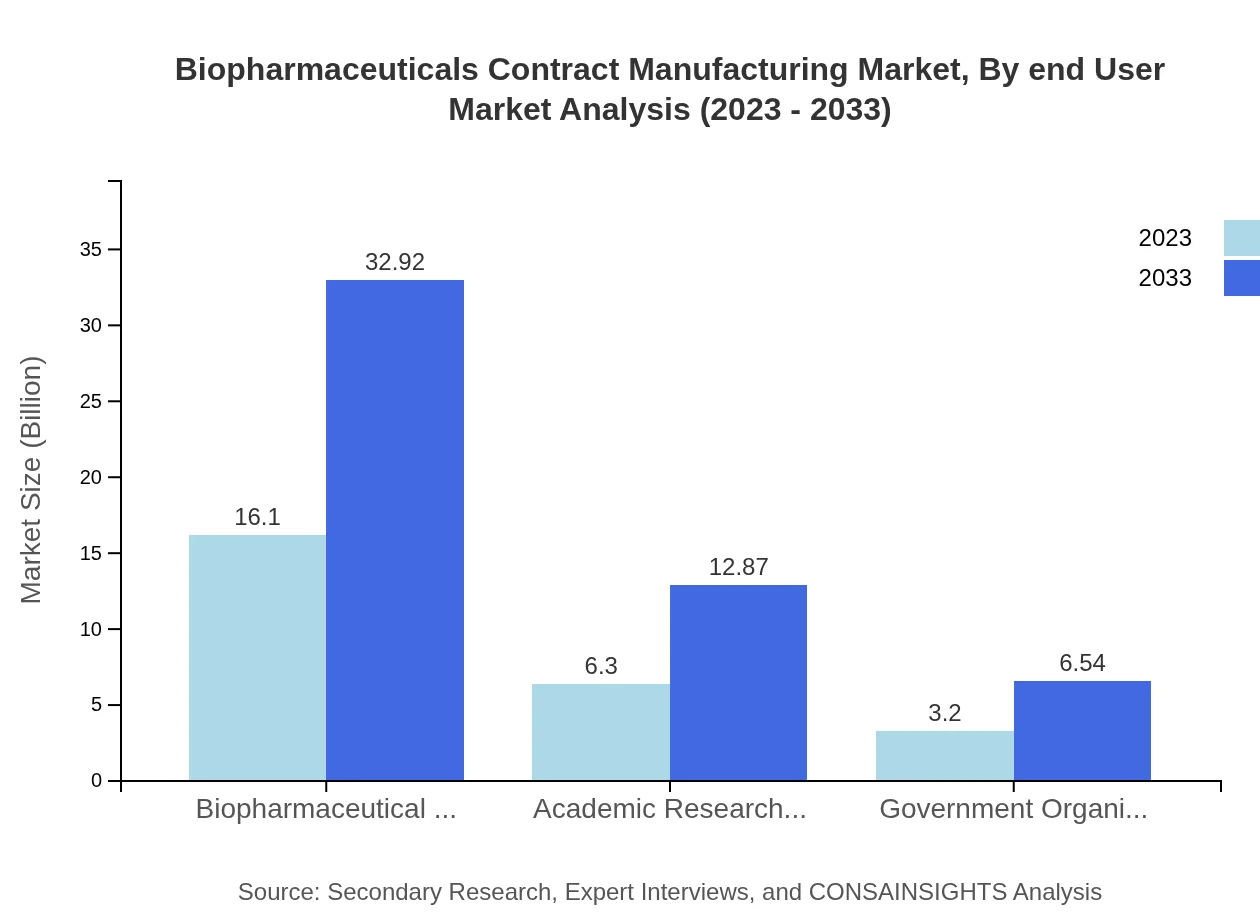
Biopharmaceuticals Contract Manufacturing Market Analysis By End User
The primary end-users include biopharmaceutical companies, academic research organizations, and government entities. Biopharmaceutical companies hold the largest market share, accounting for over 60% of the overall biopharmaceuticals contract manufacturing sector, underlining their reliance on external partners for efficiency and scalability.
Biopharmaceuticals Contract Manufacturing Market Analysis By Region Analysis
Regional analysis reveals that North America and Europe are leading in terms of market share, driven by advanced healthcare infrastructure and high R&D investments. Asia Pacific is emerging with rapid growth prospects due to increasing manufacturing capacities and strategic investment opportunities.
Biopharmaceuticals Contract Manufacturing Market Analysis By Regulatory Compliance
Compliance with FDA regulations, EMA guidelines, and current good manufacturing practices (cGMP) remains critical for market growth. Adherence to these regulations ensures product quality and safety, thus fostering consumer trust and enhancing market sustainability.
Biopharmaceuticals Contract Manufacturing Market Trends and Future Forecast
Tell us your focus area and get a customized research report.
Global Market Leaders and Top Companies in Biopharmaceuticals Contract Manufacturing Industry
Lonza Group:
Lonza is a Swiss multinational that provides integrated and sustainable solutions to healthcare and life sciences, with a strong focus on biologics contract manufacturing.Samsung Biologics:
Samsung Biologics is a South Korean company recognized for its robust contract manufacturing services, specializing in cell culture and biologics production.WuXi AppTec:
WuXi AppTec offers comprehensive services in the biopharmaceutical sector, including contract manufacturing, R&D, and regulatory advisory to support accelerated product development.We're grateful to work with incredible clients.









FAQs
What is the market size in 2023?
The market size in 2023 is $25.60 Billion. Forecasts in the report show expansion to $52.33 Billion by 2033, reflecting a 7.2% CAGR across the 2023 to 2033 period and rising outsourcing trends.
How big will the market be in 2033?
By 2033 the market is projected at $52.33 Billion, up from $25.60 Billion in 2023. This growth corresponds to a 7.2% CAGR for the 2023 to 2033 forecast period.
What is CAGR for the forecast period?
The compound annual growth rate for the 2023 to 2033 period is 7.2%. This rate underpins expansion from $25.60 Billion in 2023 to a projected $52.33 Billion by 2033.
Why is North America the largest region?
North America is the largest region due to established biopharma ecosystems, infrastructure investment, and demand for outsourced manufacturing, supporting a rise from $9.33 Billion in 2023 to $19.08 Billion in 2033.
Which companies are leading the market?
Top companies named in the report include Lonza Group, Samsung Biologics, and WuXi AppTec. These providers are noted for expanding capacity and specialized services for complex biologic manufacturing.
Who are the main end users for contract manufacturing?
Primary end users are biopharmaceutical companies, academic research organizations, and government organizations. These groups rely on CDMOs for cell culture, purification, and formulation and filling services.
What drives regional growth in Asia Pacific?
Asia Pacific grows from $4.82 Billion in 2023 to $9.84 Billion in 2033, driven by increasing biopharma activity, capacity additions, and rising demand for outsourced production across the region.
How big is the market in Europe and Latin America?
Europe expands from $7.81 Billion in 2023 to $15.97 Billion in 2033, while Latin America grows from $1.15 Billion to $2.36 Billion over the same 2023 to 2033 forecast period, reflecting regional investments.
Why is regulatory compliance emphasized in the report?
Regulatory compliance—Current Good Manufacturing Practices, FDA regulations, and EMEA guidelines—is emphasized because adherence to these standards is essential for quality, market access, and partnership selection by biopharma firms.