Acute Ischemic Stroke Diagnosis Market Report
Published Date: 21 April 2026 | Report Code: acute-ischemic-stroke-diagnosis
Acute Ischemic Stroke Diagnosis Market Size, Share, Industry Trends and Forecast to 2033
This report offers a comprehensive analysis of the Acute Ischemic Stroke Diagnosis market, including current market trends, segmentation, insights across various regions, technology advancements, and a forward-looking forecast for the years 2023 to 2033.
Key Takeaways
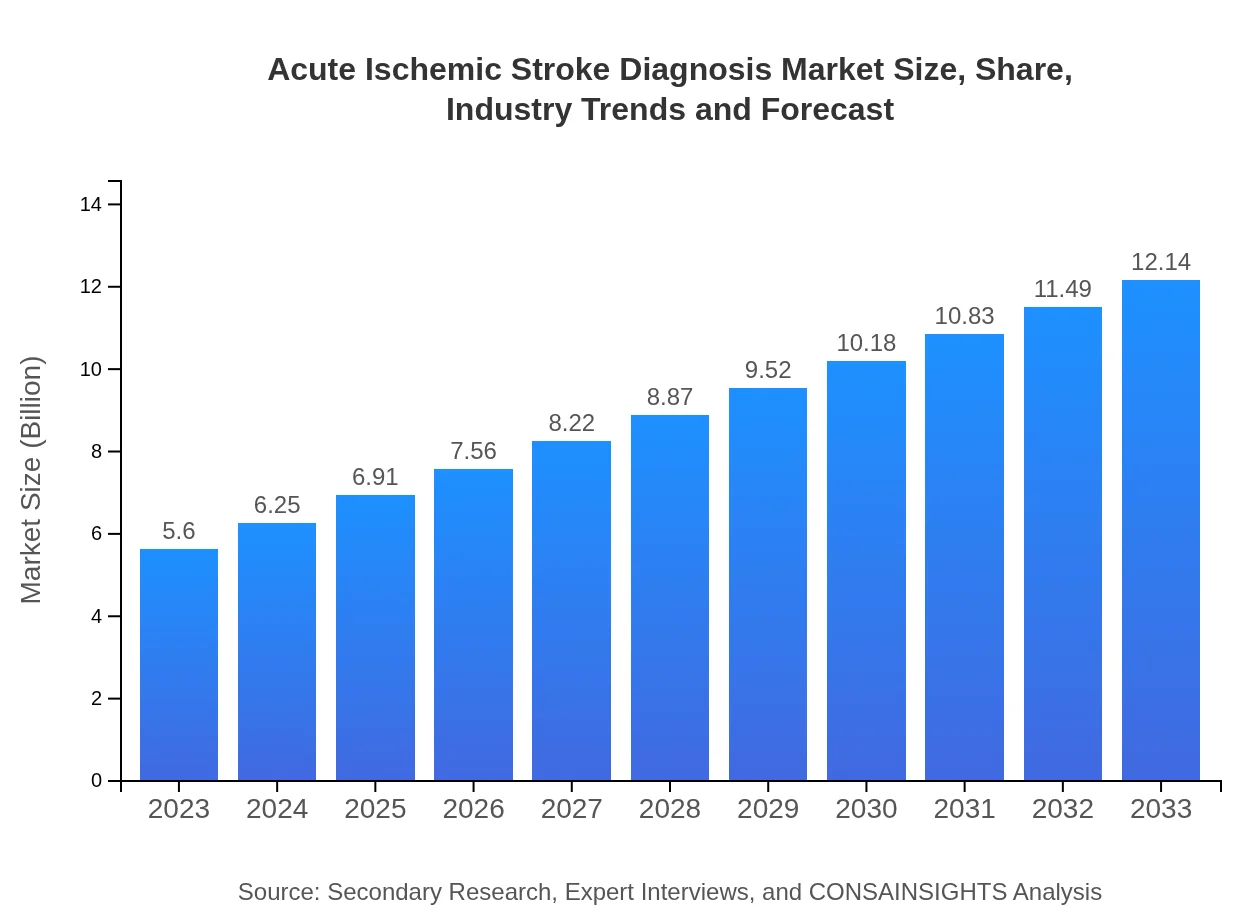
- Market expands from $5.60 Billion in 2023 to $12.14 Billion by 2033 at a 7.8% CAGR.
- North America represents the largest share and the most rapid growth among regions.
- Advanced imaging and point-of-care testing are central adoption drivers across healthcare providers.
- Hospitals, diagnostic centers, and research institutes form the primary end-user base.
- Leading vendors include GE Healthcare, Siemens Healthineers, Philips Healthcare, Abbott Laboratories, and Roche Diagnostics.
Acute Ischemic Stroke Diagnosis Market Report — Executive Summary
The report outlines the Acute Ischemic Stroke Diagnosis market’s trajectory from $5.60 Billion in 2023 to $12.14 Billion by 2033, driven by technological progress and heightened clinical emphasis on timely detection. Market expansion reflects stronger uptake of advanced imaging modalities and decentralized testing across hospitals, diagnostic centers, and research institutes. North America is identified as both the largest and fastest-growing region, supported by concentrated investment and technology deployment. Competitive dynamics are shaped by established medical device and diagnostics companies such as GE Healthcare, Siemens Healthineers, Philips Healthcare, Abbott Laboratories, and Roche Diagnostics. The study evaluates segmentation by technology, patient type, diagnosis method, and end-user, offering a structured view of adoption patterns and unmet needs. Forecasts and insights are informed by primary expert interviews and secondary sources, with attention to innovation trends and market entry considerations for stakeholders.
Key Growth Drivers
- Advances in imaging platforms and diagnostic instrumentation improving diagnostic speed and accuracy.
- Rising clinical focus on early ischemic stroke detection and intervention across care pathways.
- Expansion of point-of-care testing enabling faster decision-making in emergency and outpatient settings.
- Ongoing investment in diagnostic R&D and adoption by hospitals and diagnostic centers bolstering technology uptake.
- Concentration of market activity in North America accelerating commercialization and scale for new solutions.
| Metric | Value |
|---|---|
| Study Period | 2023 - 2033 |
| 2023 Market Size | $5.60 Billion |
| CAGR (2023-2033) | 7.8% |
| 2033 Market Size | $12.14 Billion |
| Top Companies | GE Healthcare, Siemens Healthineers, Philips Healthcare, Abbott Laboratories, Roche Diagnostics |
| Last Modified Date | 21 April 2026 |
Acute Ischemic Stroke Diagnosis Market Overview
Customize Acute Ischemic Stroke Diagnosis Market Report market research report
- ✔ Get in-depth analysis of Acute Ischemic Stroke Diagnosis market size, growth, and forecasts.
- ✔ Understand Acute Ischemic Stroke Diagnosis's regional dynamics and industry-specific trends.
- ✔ Identify potential applications, end-user demand, and growth segments in Acute Ischemic Stroke Diagnosis
What is the Market Size & CAGR of Acute Ischemic Stroke Diagnosis Market Report market in 2023?
Acute Ischemic Stroke Diagnosis Industry Analysis
Acute Ischemic Stroke Diagnosis Market Segmentation and Scope
Tell us your focus area and get a customized research report.
Acute Ischemic Stroke Diagnosis Market Report Market Analysis Report by Region
Europe Acute Ischemic Stroke Diagnosis Market Report:
Europe grows from $1.51 Billion in 2023 to $3.28 Billion in 2033. Regional expansion reflects adoption of improved diagnostic modalities and evolving clinical protocols that support broader use of imaging and point-of-care solutions.Asia Pacific Acute Ischemic Stroke Diagnosis Market Report:
Asia Pacific rises from $1.13 Billion in 2023 to $2.45 Billion in 2033. Growth is tied to increasing investment in healthcare diagnostics, gradual uptake of advanced technologies, and expanding services in hospitals and diagnostic centers.North America Acute Ischemic Stroke Diagnosis Market Report:
North America increases from $2.08 Billion in 2023 to $4.5 Billion in 2033. As the largest and fastest-growing region, it benefits from concentrated technology adoption, strong clinical infrastructure, and investment that accelerate deployment of advanced diagnostic platforms.South America Acute Ischemic Stroke Diagnosis Market Report:
Middle East & Africa Acute Ischemic Stroke Diagnosis Market Report:
Middle East and Africa increase from $0.42 Billion in 2023 to $0.91 Billion in 2033. Development is supported by incremental investments in clinical diagnostics and selective adoption of imaging and laboratory-based approaches across care settings.Tell us your focus area and get a customized research report.
Research Methodology
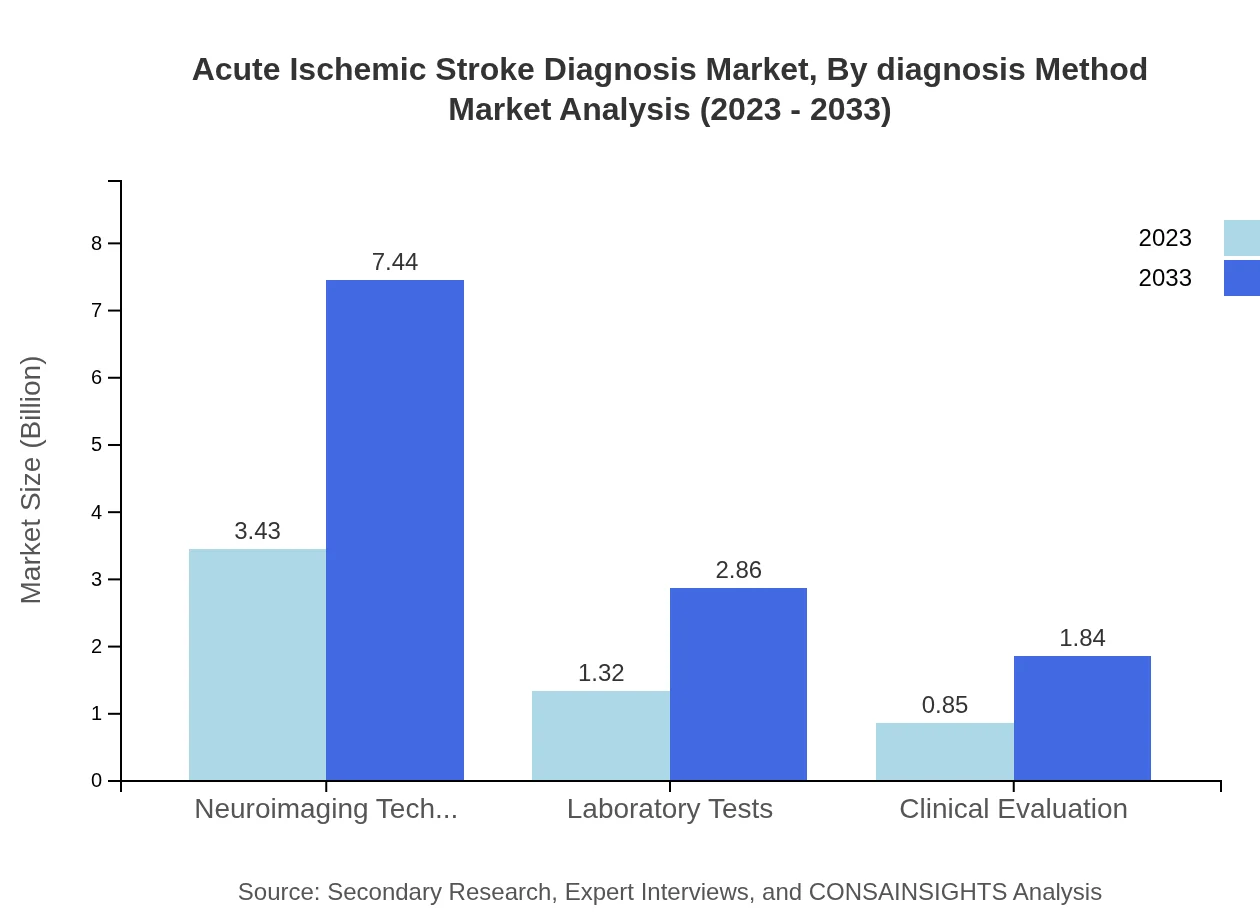
Acute Ischemic Stroke Diagnosis Market Analysis By Diagnosis Method
Neuroimaging techniques, including advanced CT and MRI scans, dominate the market with a substantial share estimated at 61.25% in 2023, set to maintain this level through 2033. Laboratory tests and clinical evaluations also contribute significantly but at relatively lower shares. The emphasis on advanced imaging technologies is indicative of the market's direction towards precision medicine.
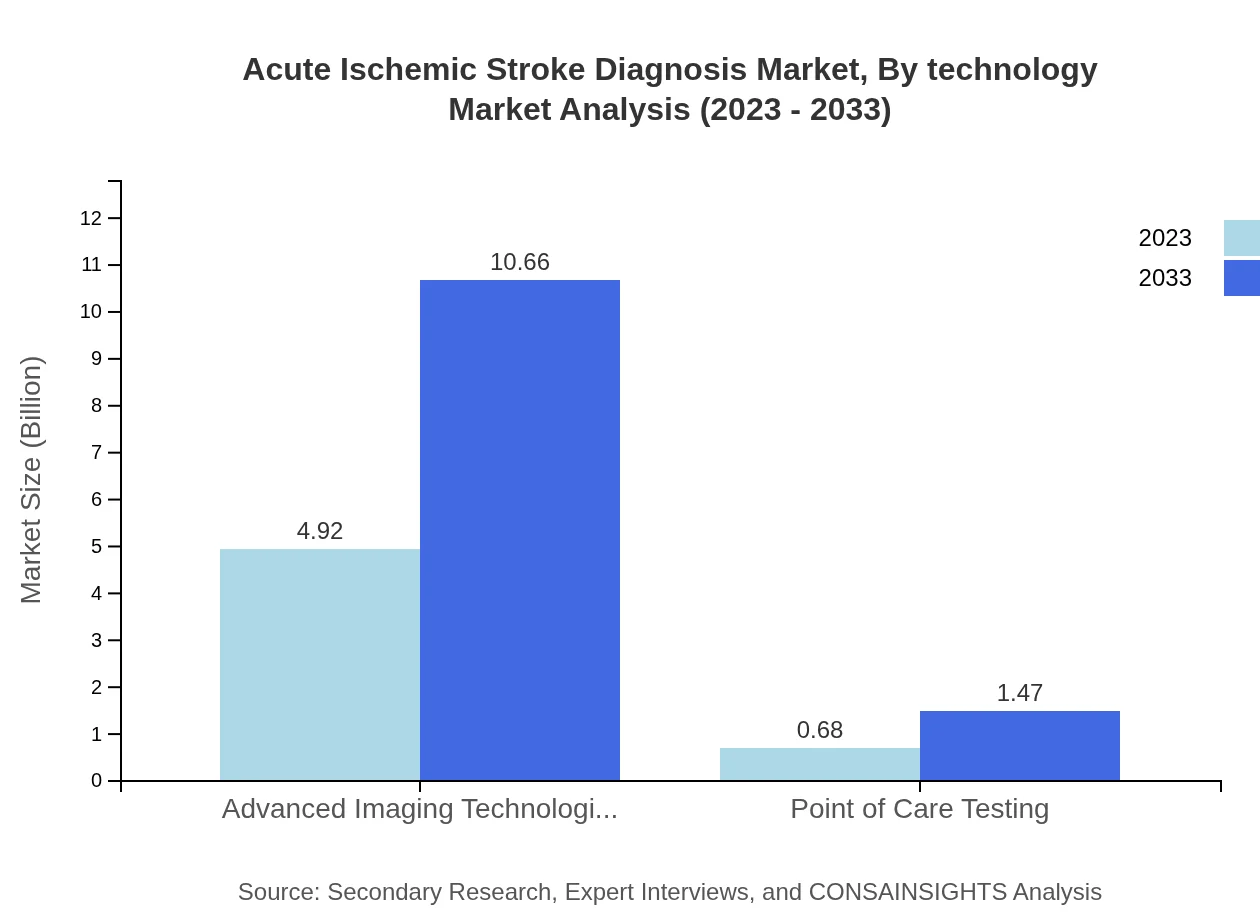
Acute Ischemic Stroke Diagnosis Market Analysis By Technology
Advanced imaging technologies are anticipated to hold an 87.85% market share in 2023, with growth expected as new modalities are developed. Point of care testing, although smaller in scale, is crucial for immediate care settings, projected to grow from 0.68 billion to 1.47 billion through 2033, reflecting advances in rapid diagnostic protocols.
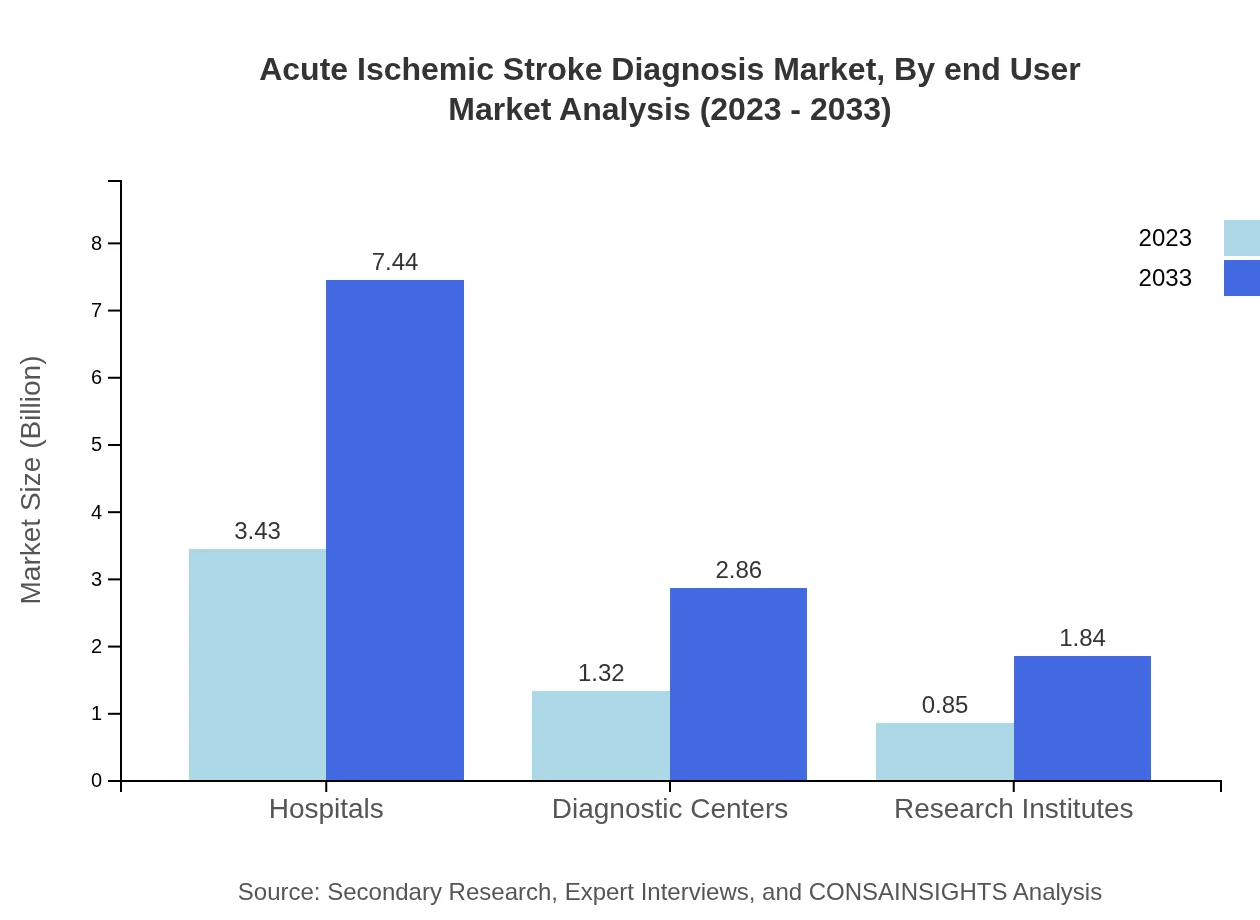
Acute Ischemic Stroke Diagnosis Market Analysis By End User
Hospitals comprise the largest segment in the Acute Ischemic Stroke Diagnosis market, accounting for 61.25% of the market share in 2023, projected to grow to 61.25% by 2033. Diagnostic centers and research institutions are crucial as they focus on developing new diagnostic methodologies, sharing 23.58% and 15.17% of the market respectively.
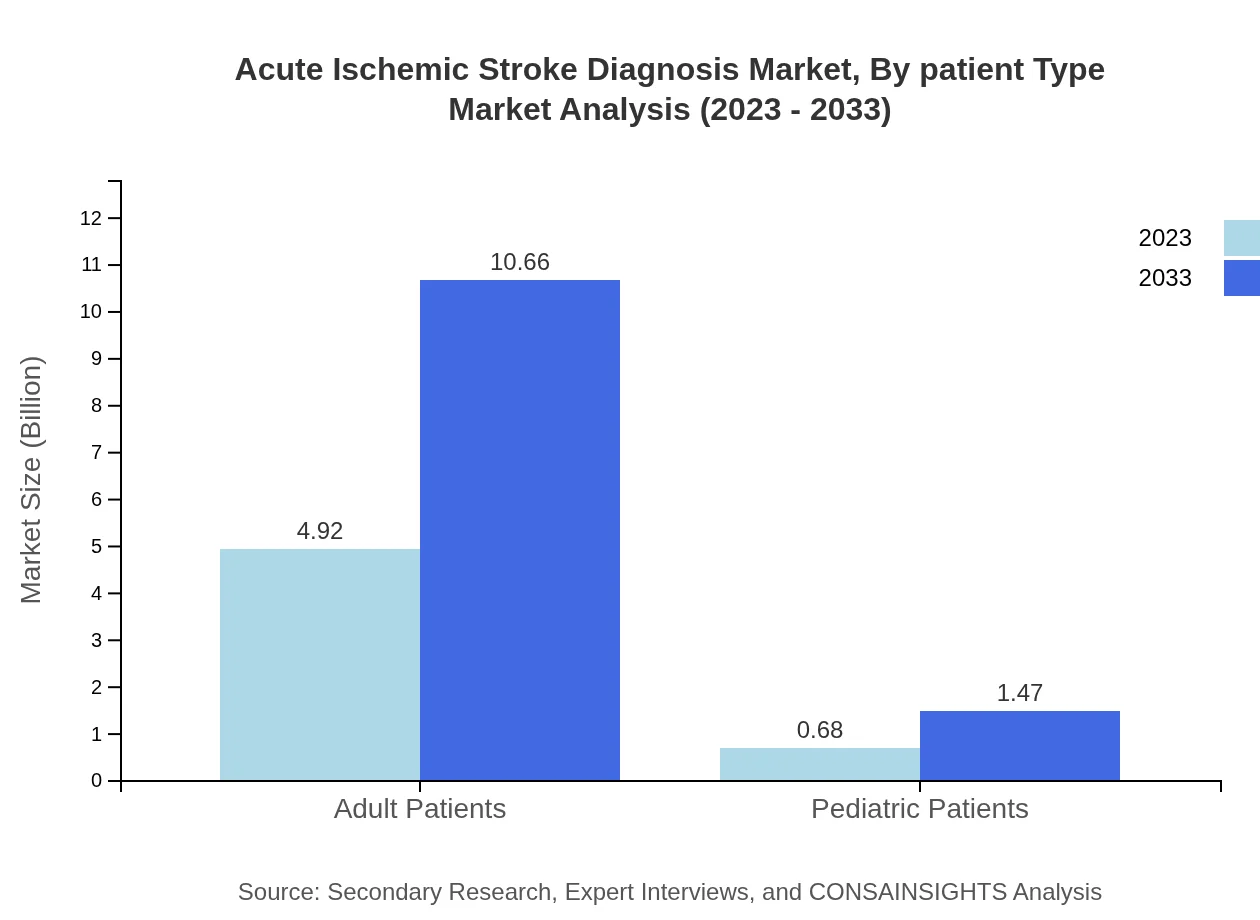
Acute Ischemic Stroke Diagnosis Market Analysis By Patient Type
Adult patients represent the majority share in the market at 87.85% in 2023, projected to remain stable through 2033. Pediatric patients, while smaller at 12.15%, show significant growth potential as awareness improves among caregivers and healthcare providers.
Acute Ischemic Stroke Diagnosis Market Trends and Future Forecast
Tell us your focus area and get a customized research report.
Global Market Leaders and Top Companies in Acute Ischemic Stroke Diagnosis Industry
GE Healthcare:
GE Healthcare is a leading innovator in medical imaging technologies and healthcare solutions. Their advancements have significantly impacted stroke diagnostics and treatment.Siemens Healthineers:
Siemens Healthineers is renowned for developing cutting-edge tools for radiology and medical imaging, contributing to faster and more accurate stroke diagnoses.Philips Healthcare:
Philips Healthcare provides advanced diagnostic imaging solutions and has been pivotal in enhancing stroke management through innovative technologies.Abbott Laboratories:
Abbott Laboratories is focused on developing efficient diagnostic tests that provide quick results, which are critical in stroke settings.Roche Diagnostics:
Roche Diagnostics specializes in diagnostic tests and is instrumental in developing new biomarkers for stroke detection, driving innovation in the field.We're grateful to work with incredible clients.









FAQs
What is the market size of the Acute Ischemic Stroke Diagnosis market in 2023?
The market size for 2023 is $5.60 Billion, as stated in the report. This figure reflects current diagnostic modality adoption across hospitals, diagnostic centers, and research institutes.
How big will the market be in 2033?
By 2033 the market is projected to reach $12.14 Billion, based on the report’s forecast covering technology adoption and expanded use across primary end users over the 2023–2033 period.
What is CAGR for the forecast period?
The reported compound annual growth rate (CAGR) for the forecast period 2023 to 2033 is 7.8%, driving the increase from $5.60 Billion to $12.14 Billion.
Who are the top companies in this market?
Key market participants listed include GE Healthcare, Siemens Healthineers, Philips Healthcare, Abbott Laboratories, and Roche Diagnostics, representing major suppliers of imaging and diagnostic tools.
What are the primary end Users for these diagnostics?
Primary end-users comprise hospitals, diagnostic centers, and research institutes, reflecting settings where acute ischemic stroke diagnosis technologies are commonly deployed for patient evaluation and care.
Which technologies are prominent in the market?
Reported technology subsegments include Advanced Imaging Technologies and Point of Care Testing, which are highlighted as central modalities for ischemic stroke diagnosis and rapid clinical decision-making.
Why is North America significant in this market?
North America is identified as both the largest and fastest-growing region, driven by concentrated technology adoption, clinical infrastructure, and investment that support broader deployment of diagnostic solutions.
What diagnosis methods are covered in the segmentation?
Segmentation by diagnosis method includes Neuroimaging Techniques, Laboratory Tests, and Clinical Evaluation, indicating a mix of imaging, biochemical, and clinical approaches used in practice.
How big is the European market in 2023 and 2033?
Europe is shown as growing from $1.51 Billion in 2023 to $3.28 Billion in 2033, reflecting regional uptake of diagnostic technologies and evolving clinical workflows over the forecast period.
What research approach was used to prepare this report?
The study combined primary interviews with industry experts and secondary research from company reports and publications, followed by data triangulation, internal validation, and expert-led trend analysis.