Biologics Safety Testing Market Report
Published Date: 22 April 2026 | Report Code: biologics-safety-testing
Biologics Safety Testing Market Size, Share, Industry Trends and Forecast to 2033
This report provides a comprehensive analysis of the Biologics Safety Testing market, covering market trends, sizes, forecasts up to 2033, and insights into segments and regional performance.
Key Takeaways
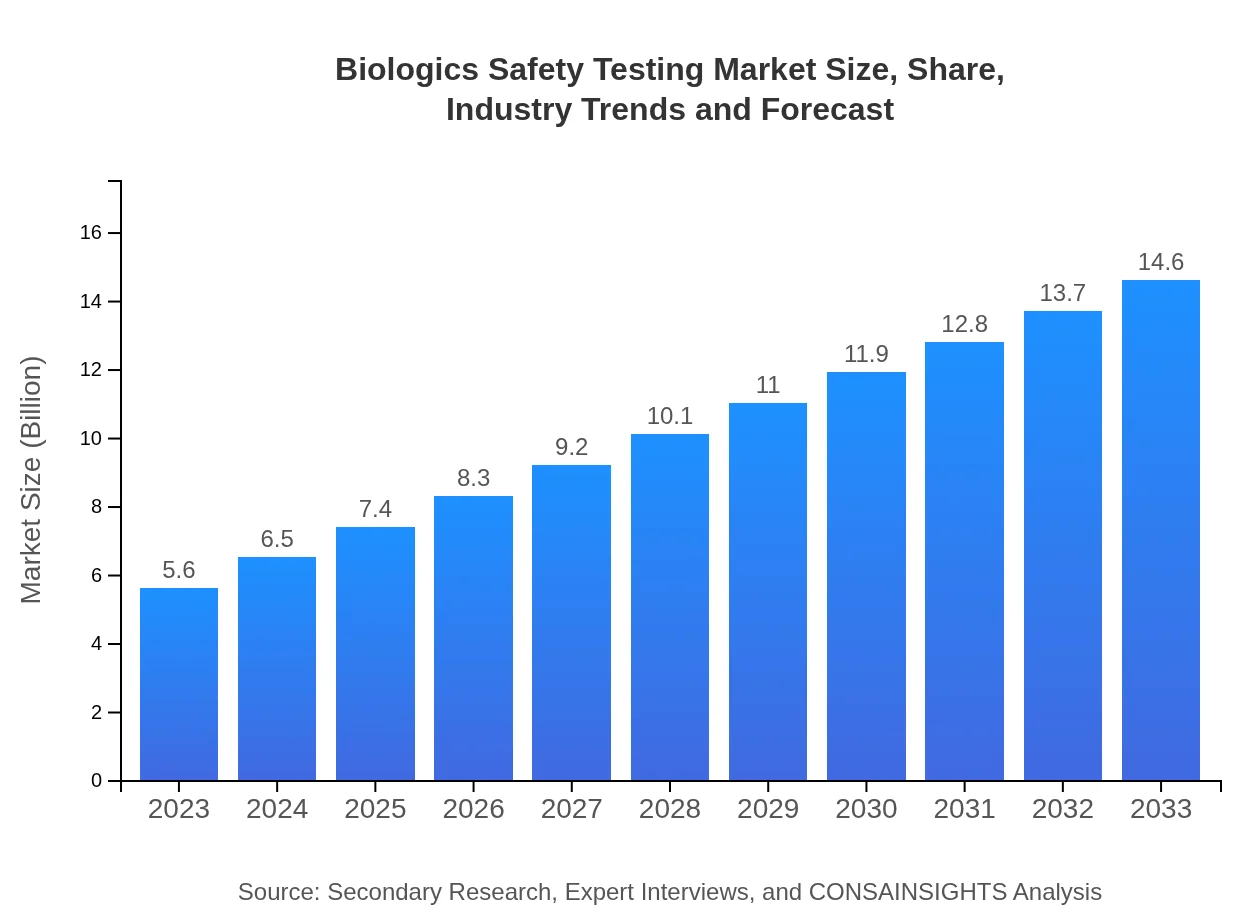
- Market expands from $5.60 Billion in 2023 to $14.60 Billion in 2033 at a 9.7% CAGR, reflecting sustained growth potential.
- North America is both the largest and fastest-growing region, outpacing other markets in adoption and investment.
- Europe rises from $1.78 Billion to $4.64 Billion, while Asia Pacific grows from $1.04 Billion to $2.73 Billion over the decade.
- Service demand centers on vaccines and therapeutics testing, with testing and consulting services in high requirement.
- Major providers such as Charles River Laboratories, Eurofins Scientific, SGS SA and LabCorp are prominent service partners.
Biologics Safety Testing Market Report — Executive Summary
This report captures the market dynamics behind rising demand for biologics safety assessment, driven by stricter regulatory oversight and expanding biologic pipelines. From a $5.60 Billion base in 2023 to $14.60 Billion by 2033, the sector is forecast to grow at a 9.7% CAGR over the 2023 to 2033 period. Investment in advanced molecular assays, in vitro models and automated platforms is reshaping service portfolios, while pharmaceutical and biotech companies increasingly outsource testing to specialized providers. Regional expansion is led by North America, supported by established infrastructure and active clinical programs. Europe and Asia Pacific also show notable growth, reflecting regulatory harmonization and rising local manufacturing. Competitive activity centers on service differentiation, strategic partnerships and capability expansion by firms such as Charles River Laboratories, Eurofins Scientific, SGS SA and LabCorp. The market structure spans methodology, product type, service offerings and end-users, with testing services and consulting forming the primary commercial models.
Key Growth Drivers
- Heightened regulatory scrutiny requiring comprehensive safety dossiers increases demand for specialized testing services.
- Rising biologics development—vaccines and therapeutics—drives need for expanded preclinical and lot-release testing.
- Technological advances in molecular assays and in vitro platforms enable more reliable, scalable safety assessments.
- Pharmaceutical and biotech outsourcing of complex testing to specialized laboratories expands service volumes and market reach.
- Strategic investments and partnerships among service providers broaden capabilities and geographic coverage.
| Metric | Value |
|---|---|
| Study Period | 2023 - 2033 |
| 2023 Market Size | $5.60 Billion |
| CAGR (2023-2033) | 9.7% |
| 2033 Market Size | $14.60 Billion |
| Top Companies | Charles River Laboratories, Eurofins Scientific, SGS SA, LabCorp |
| Last Modified Date | 22 April 2026 |
Biologics Safety Testing Market Overview
Customize Biologics Safety Testing Market Report market research report
- ✔ Get in-depth analysis of Biologics Safety Testing market size, growth, and forecasts.
- ✔ Understand Biologics Safety Testing's regional dynamics and industry-specific trends.
- ✔ Identify potential applications, end-user demand, and growth segments in Biologics Safety Testing
What is the Market Size & CAGR of Biologics Safety Testing Market Report market in 2023?
Biologics Safety Testing Industry Analysis
Biologics Safety Testing Market Segmentation and Scope
Tell us your focus area and get a customized research report.
Biologics Safety Testing Market Report Market Analysis Report by Region
Europe Biologics Safety Testing Market Report:
Europe grows from $1.78 Billion in 2023 to $4.64 Billion in 2033. Regional expansion is driven by regulatory harmonization, strong contract research and testing infrastructures, and increasing biologics development across multiple therapeutic areas.Asia Pacific Biologics Safety Testing Market Report:
Asia Pacific increases from $1.04 Billion in 2023 to $2.73 Billion in 2033. Rising local biologics manufacturing, expanding clinical programs and investments in laboratory capabilities support market acceleration across the region.North America Biologics Safety Testing Market Report:
North America is the largest and fastest-growing region, expanding from $1.97 Billion in 2023 to $5.13 Billion in 2033. Growth is supported by concentrated pharma and biotech activity, established regulatory systems, and high adoption of advanced testing technologies and outsourced laboratory services.South America Biologics Safety Testing Market Report:
Middle East & Africa Biologics Safety Testing Market Report:
Middle East and Africa expands from $0.45 Billion in 2023 to $1.18 Billion in 2033. Market progression is influenced by growing healthcare investments, regional regulatory development and rising need for outsourced safety testing services.Tell us your focus area and get a customized research report.
Research Methodology
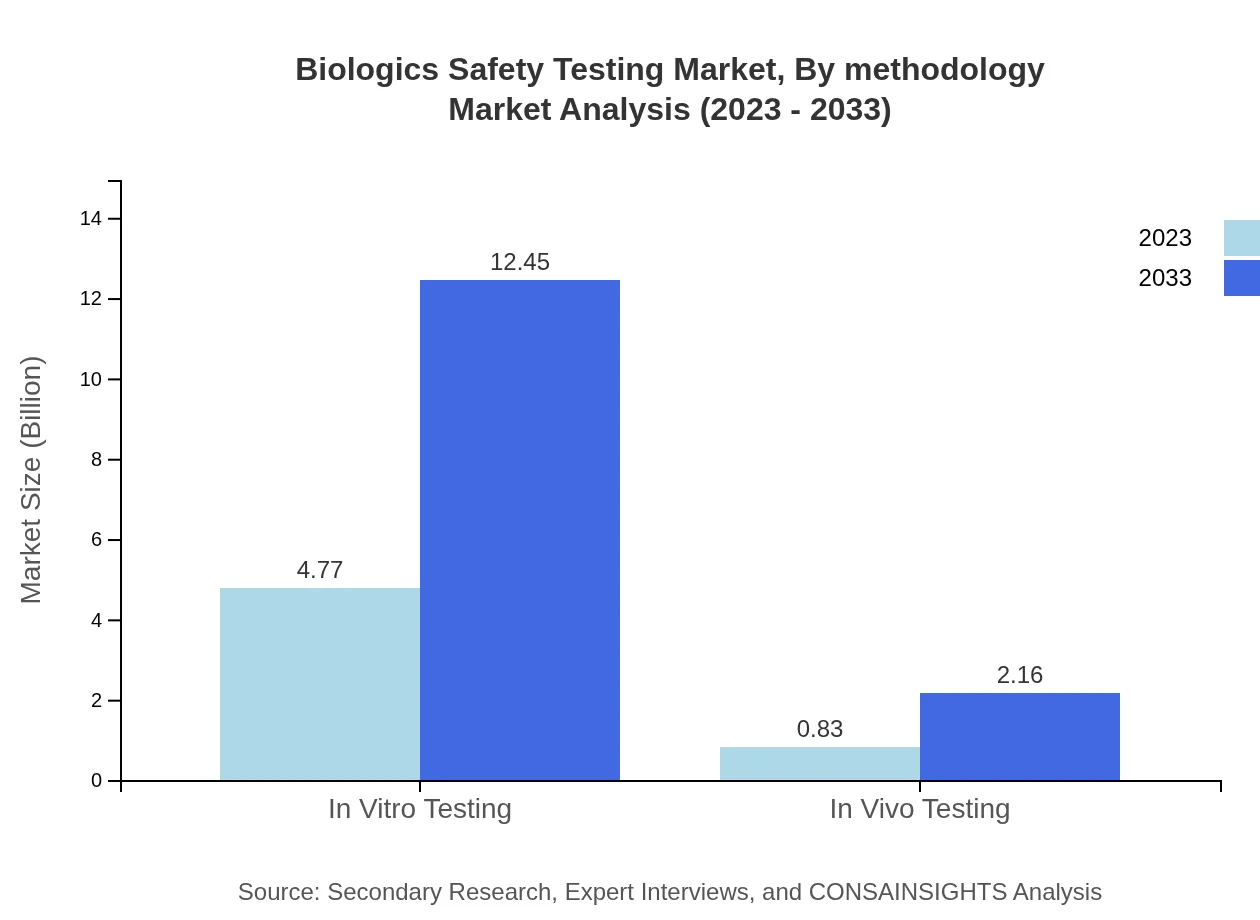
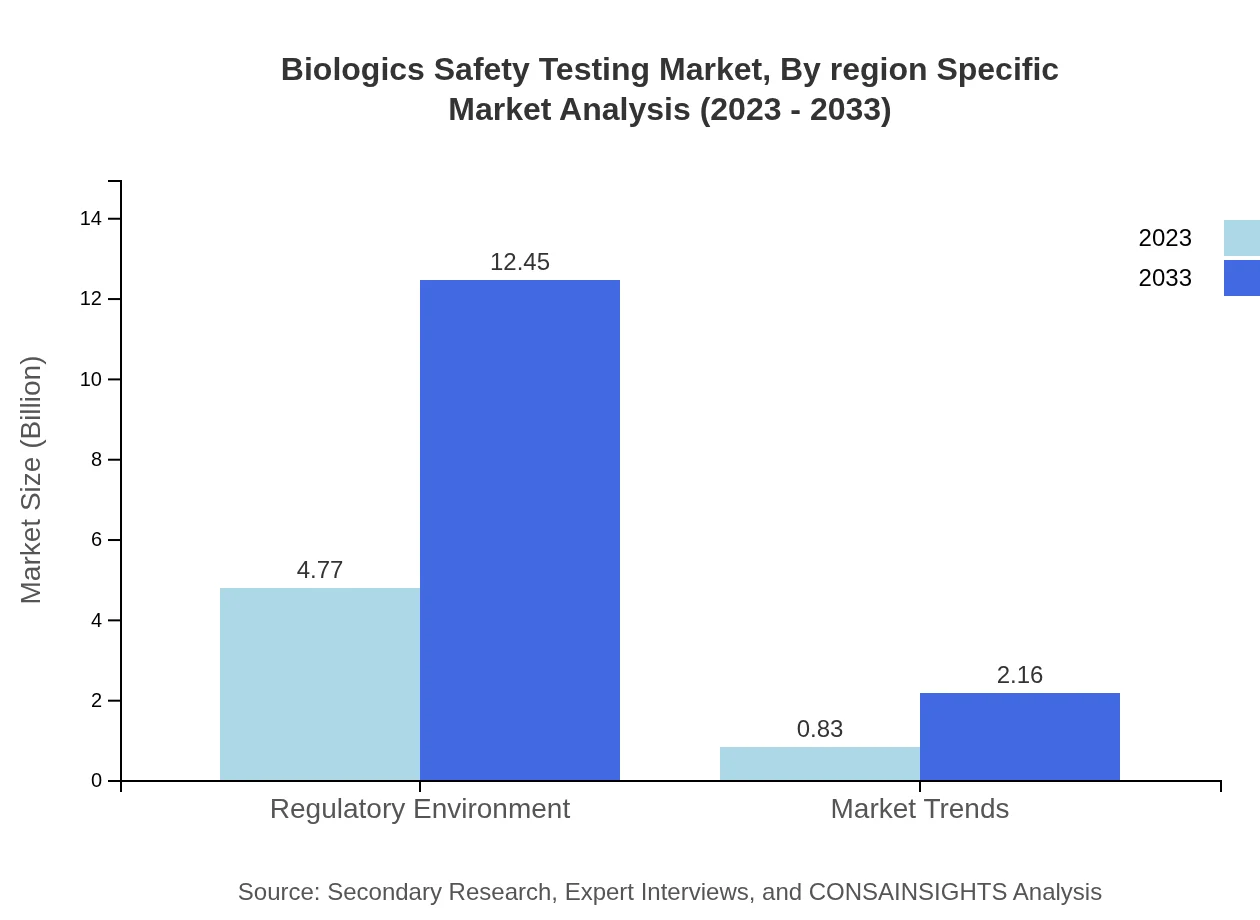
Biologics Safety Testing Market Analysis By Methodology
The segment analysis highlights a significant reliance on in vitro testing, which dominates the market with a size of $4.77 billion in 2023, expected to grow to $12.45 billion by 2033. In vivo testing, though smaller, shows substantial growth potential, expanding from $0.83 billion to $2.16 billion in the same period.
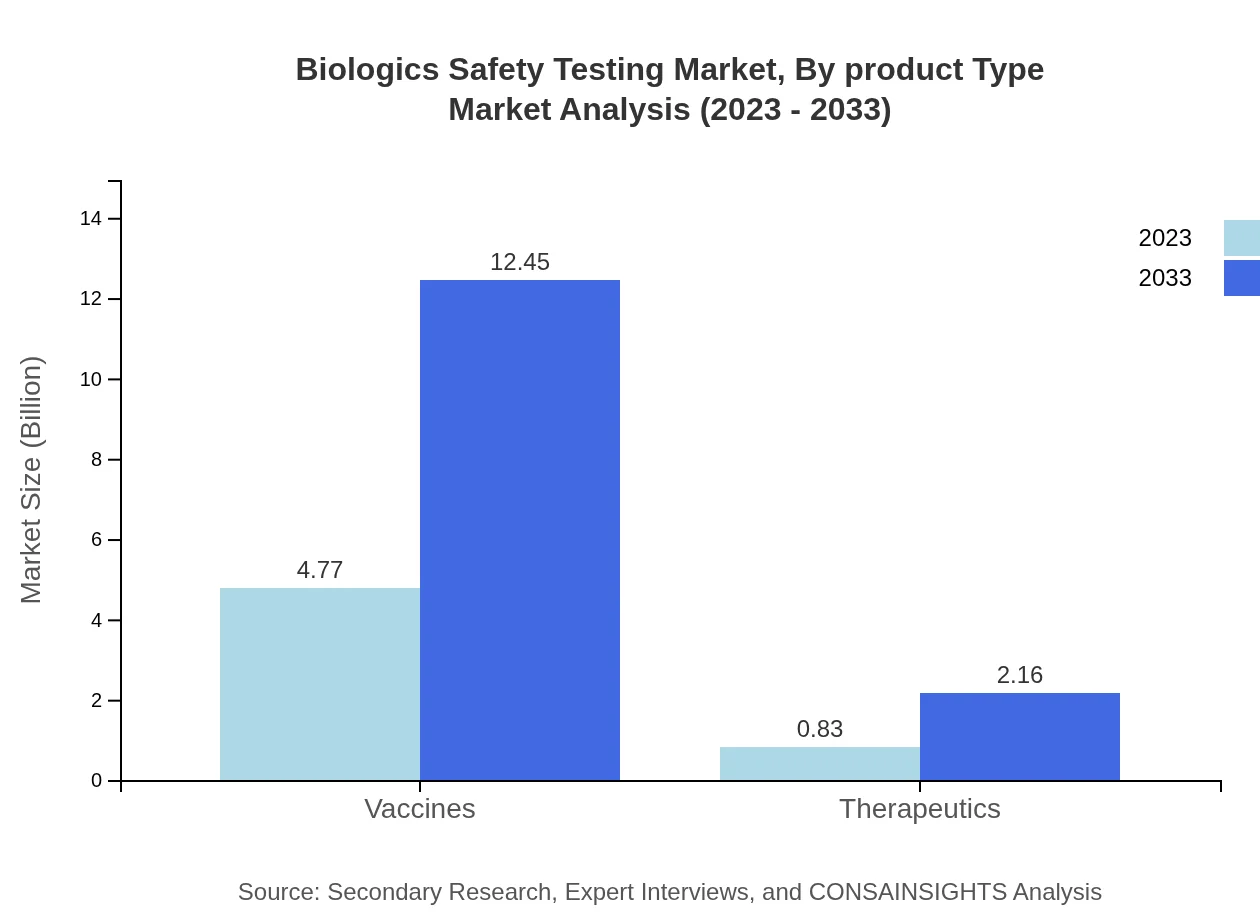
Biologics Safety Testing Market Analysis By Product Type
Vaccines make up a substantial portion of the market, growing from $4.77 billion in 2023 to $12.45 billion by 2033. Therapeutics and diagnostics are also critical, showing a rising demand as they play a vital role in compliance with safety standards.
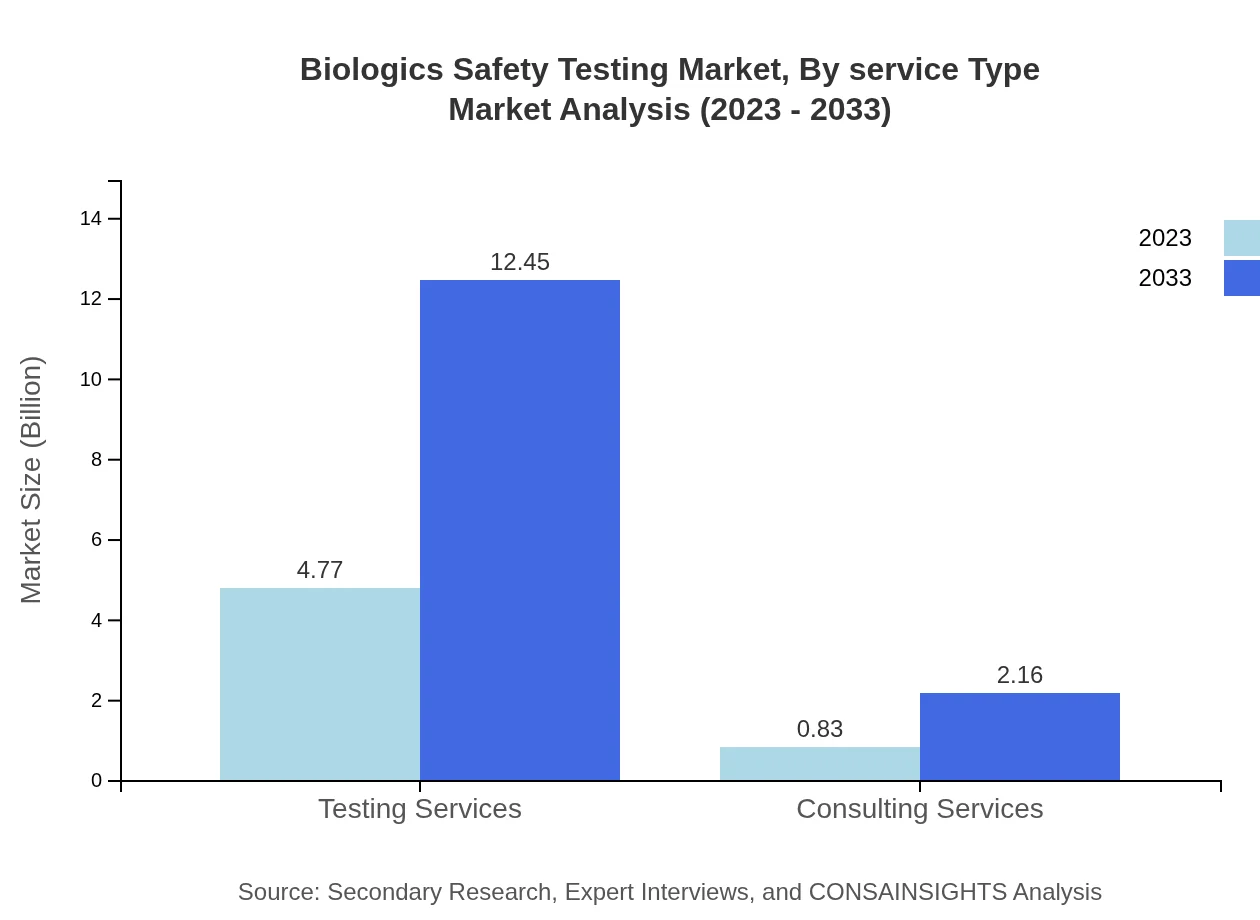
Biologics Safety Testing Market Analysis By Service Type
Testing services encompass the majority of the market share, with significant contributions from consulting services. Testing services are set to increase from $4.77 billion in 2023 to $12.45 billion in 2033, asserting their importance in the industry.
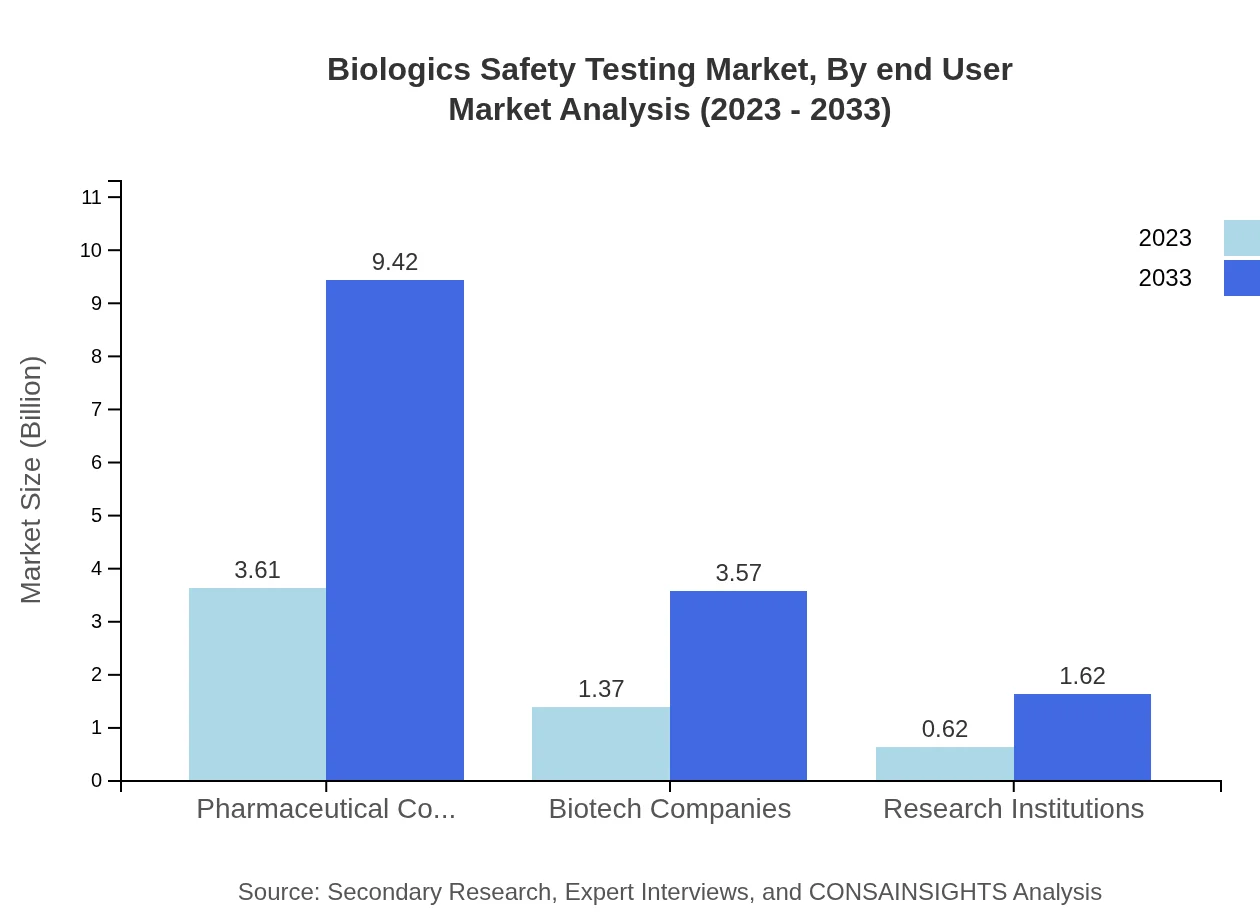
Biologics Safety Testing Market Analysis By End User
Pharmaceutical and biotech companies lead the end-user segment, with a market size of $3.61 billion expected to reach $9.42 billion by 2033. Research institutions also play a significant role, with revenues projected to rise from $0.62 billion to $1.62 billion.
Biologics Safety Testing Market Analysis By Region Specific
Various regional factors uniquely influence market growth. North America drives innovation due to mature infrastructure, while Asia Pacific benefits from rapid expansion in production capabilities. Regulatory challenges in Europe also indicate essential regional comparisons.
Biologics Safety Testing Market Trends and Future Forecast
Tell us your focus area and get a customized research report.
Global Market Leaders and Top Companies in Biologics Safety Testing Industry
Charles River Laboratories:
A leading provider of preclinical and clinical laboratory services for the biopharmaceutical industry, Charles River is essential in providing comprehensive safety testing services.Eurofins Scientific:
Specializing in bioanalytical testing, Eurofins offers a range of services necessary for biologics safety testing, helping pharmaceutical companies ensure their products meet regulatory compliance.SGS SA:
With a global presence, SGS provides inspection, verification, testing, and certification services, including crucial testing for biologics to ensure compliance and safety.LabCorp:
LabCorp plays a significant role in laboratory testing and provides comprehensive solutions for biologics safety assessment, supporting the development pipeline of many medications.We're grateful to work with incredible clients.









FAQs
What is the current market size in 2023?
The market size in 2023 is $5.60 Billion. This figure serves as the reported baseline for the forecast, reflecting the combined revenue of testing services, methodologies and related offerings in that year.
How big will the market be in 2033?
By 2033 the market is reported at $14.60 Billion. That end-value represents projected growth across services, methodologies and regional expansion over the 2023 to 2033 forecast period.
What is CAGR for the forecast period?
The compound annual growth rate for the 2023 to 2033 forecast period is 9.7%. This reflects the aggregate annualized growth expected across regions and service segments.
Why is North America the largest and fastest Growing region?
North America leads due to established regulatory frameworks, dense biotech and pharmaceutical activity, high outsourcing demand and advanced laboratory infrastructure supporting extensive safety testing programs.
Which regions show notable expansion between 2023 and 2033?
Europe, Asia Pacific, Latin America and Middle East & Africa all register growth. Europe rises from $1.78 Billion to $4.64 Billion while Asia Pacific moves from $1.04 Billion to $2.73 Billion.
Who are the leading companies in this market?
Prominent service providers include Charles River Laboratories, Eurofins Scientific, SGS SA and LabCorp, which offer broad testing portfolios and geographic coverage for biologics safety assessments.
What drives demand for testing services?
Demand is propelled by increasing biologic product pipelines, stricter regulatory requirements, and a shift toward outsourcing complex assays to specialized laboratories with validated methodologies.
What methodologies are prominent in biologics safety testing?
Key methodologies include in vitro testing and in vivo testing. These approaches are widely used across preclinical and quality control workflows to evaluate biologic safety and consistency.